Reports
Reports
Within the healthcare sector, reprocessing of ‘single-use’ devices has become mainstream across hospitals and clinics as a measure to reduce costs. However, while reprocessing enables healthcare providers to reduce costs, there is a downside to it– the risk of cross-contamination and pathogen infection. The number of hospital-acquired infections (HAI) has increased at a steady pace across the global healthcare sector and thus, medical device cleaning processes have garnered significant attention from regulatory bodies. At present, medical device manufacturers are increasingly focusing on providing robust guidelines and manuals that offer insights regarding sterilization, disinfection, and cleaning of ‘single-use’ medical devices.
Thus, medical device cleaning validations have become an integral part of the reprocessing validation cycle. Moreover, it also assures that medical devices can be cleaned and sterilized by adhering to the medical device manufacturer’s instruction for use (IFU).
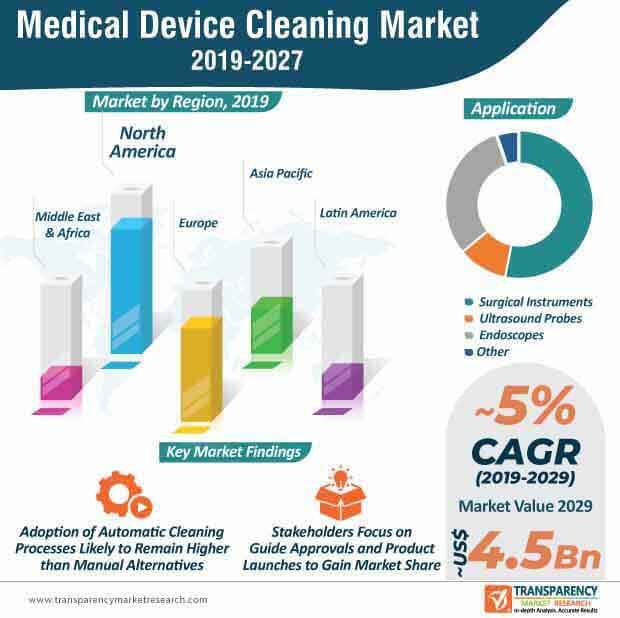
While the prevalence of hospital acquired infections continues to grow across ambulatory clinics, surgical centers, hospitals, etc., the medical device cleaning market has also gained notable momentum. In the current scenario, in the U.S., guidelines provided by The American Society of Testing and Materials (ASTM) related to medical device cleaning are applicable, which predominantly focus on equipment design, documentation, sampling, analytical techniques, procedures, etc. Owing to these developments in the healthcare sector concerning medical device cleaning, the medical device cleaning market is projected to reach a value of ~US$ 3.2 Bn by the end of 2020.
Scientific advancements in therapeutic and diagnostic medicine have triggered the production of cutting-edge reusable medical devices. However, due to the risk of cross-contamination and a range of other infections, medical device manufacturers have the responsibility to validate their product label claims as far as reusability is concerned. Label guidelines require approval from the Food and Drug Association (FDA) to ensure the overall efficacy and safety of the provided guidelines. The efficacy of the guidelines primarily depends on the markers utilized to soil the devices prior to cleaning. Some of the most commonly used markers include hemoglobin and protein.
Medical device cleaning techniques have evolved over the years and are one of the most critical stages in the reprocessing of reusable medical devices. Cleaning techniques are primarily segregated into manual and automatic processes. Manual medical device cleaning processes are largely deployed to clean highly complex and delicate medical devices, such as lensed instruments, microsurgical devices, and flexible endoscopes among others. Automated cleaning techniques are predominantly used when other cleaning techniques prove to be ineffective, to prevent soiling of crevices, joints, lumens, and other parts of a medical device. Within the medical device cleaning market, the automatic cleaning process segment is estimated to reach a value of ~US$ 2 Bn and account for ~63% share of the medical device cleaning market by the end of 2020. The automatic cleaning process segment is also projected to expand at a higher CAGR than manual cleaning process segment during the forecast period (2019-2027).
The American Society of Testing and Material is an important regulatory body that is responsible for the approval of new standards for medical device cleaning. Testing laboratories, regulatory agencies, and medical device manufacturers are required to comply with these standards. In the medical device cleaning market, attaining approval from the ASTM for new standards has gained significant momentum in recent times.
For instance, in November 2019, the ASTM approved a list of new standards wherein the effectiveness of new medical device cleaning procedures is thoroughly evaluated. Additionally, the newly curated guide will largely focus on the extraction process of test soils to verify new cleaning methods. According to a member of the ATSM, approval of new standards is a crucial step in the overall process of validating cleaning instructions.
Stakeholders in the medical device cleaning market are also focusing their efforts on the launch of new medical device cleaning products. For instance, in April 2019, DuPont launched a medical device cleaning bio-based enzyme – OPTIMASE. The product, which is part of the company’s new product range is primarily developed to provide optimum cleaning efficiency in medical devices.

Analysts’ Viewpoint
The medical device cleaning market is estimated to grow at a CAGR of 5.2% during the forecast period. A significant rise in the number of hospital-acquired infections is a leading factor that has increased brought medical device cleaning at the forefront in the past couple of decades. Moreover, as medical device manufacturers are required to provide guidelines and standards for cleaning, sterilization, etc., the market for medical device cleaning is expected to witness significant growth in the coming years. Automatic cleaning processes will remain popular during the forecast period despite steady adoption of manual cleaning processes. Product launches and mergers & acquisitions are likely to become common in the medical device cleaning market landscape.
Medical device cleaning market to reach A valuation of US$ 4.53 Bn by 2027
Medical device cleaning market is expected to expand at a CAGR of 5.2% from 2019 to 2027
Medical device cleaning market is driven by increase in the number of surgeries and rise in the demand for endoscopic procedures in emerging economies across the globe
Asia Pacific accounted for a major share of the global medical device cleaning market , owing to a large number of patients with chronic burn injuries and cancer
Key players operating in the global medical device cleaning market include 3M Company, Getinge AB, Cantel Medical Corp., STERIS plc., Fortive Corporation, Oro Clean Chemie AG
1. Preface
1.1. Report Scope and Market Segmentation
1.2. Research Highlights
2. Assumptions and Research Methodology
2.1. Assumptions and Acronyms Used
2.2. Research Methodology
3. Executive Summary
3.1. Global Medical Device Cleaning Market: Market Snapshot
4. Market Overview
4.1. Global Medical Device Cleaning Market: Type Overview
4.2. Global Medical Device Cleaning Market: Key Industry Developments
4.3. Market Dynamics
4.4. Drivers and Restraints Snapshot Analysis
4.5. Drivers
4.5.1. Surge in number of surgeries
4.5.2. Aging global population
4.6. Restraints
4.6.1. Shift in trend toward utilization of disposable medical devices
4.7. Opportunities
4.8. Global Medical Device Cleaning Market Revenue Projections (US$ Mn), 2017–2027
5. Key Insights
5.1. Key Mergers & Acquisitions
5.2. Guidelines for Disinfection and Sterilization in Healthcare
5.3. Top 3 players operating in the market
6. Global Medical Device Cleaning Market Analysis, by Product
6.1. Introduction
6.2. Global Medical Device Cleaning Market Value and Forecast, by Product
6.2.1. Chemicals
6.2.1.1. Aldehydes
6.2.1.2. Hydrogen Peroxide
6.2.1.3. Alcohol
6.2.1.4. Chlorine based Disinfectant
6.2.1.5. Peracetic Acid
6.2.1.6. Quaternary Ammonium Compounds
6.2.1.7. Others
6.2.2. Detergents
6.3. Global Medical Device Cleaning Market Analysis, by Product
6.4. Global Medical Device Cleaning Market Attractiveness Analysis, by Product
7. Global Medical Device Cleaning Market Analysis, by Process
7.1. Introduction
7.2. Global Medical Device Cleaning Market Value and Forecast, by Process
7.2.1. Manual Cleaning
7.2.2. Automatic Cleaning
7.3. Global Medical Device Cleaning Market Analysis, by Process
7.4. Global Medical Device Cleaning Market Attractiveness Analysis, by Process
8. Global Medical Device Cleaning Market Analysis, by Application
8.1. Introduction
8.2. Global Medical Device Cleaning Market Value and Forecast, by Application
8.2.1. Surgical Instruments
8.2.2. Ultrasound Probes
8.2.3. Endoscopes
8.2.4. Others
8.3. Global Medical Device Cleaning Market Analysis, by Application
8.4. Global Medical Device Cleaning Market Attractiveness Analysis, by Application
9. Global Medical Device Cleaning Market Analysis, by Region
9.1. Global Medical Device Cleaning Market Value and Forecast, by Region, 2017–2027
9.1.1. North America
9.1.2. Europe
9.1.3. Japan
9.1.4. Rest of the World
9.2. Global Medical Device Cleaning Market Analysis, by Region
9.3. Global Medical Device Cleaning Market Attractiveness Analysis, by Region
10. North America Medical Device Cleaning Market Analysis
10.1. North America Medical Device Cleaning Market
10.2. North America Medical Device Cleaning Market Overview
10.3. North America Medical Device Cleaning Market Value and Forecast, by Product
10.3.1. Chemicals
10.3.1.1. Aldehydes
10.3.1.2. Hydrogen Peroxide
10.3.1.3. Alcohol
10.3.1.4. Chlorine based Disinfectant
10.3.1.5. Peracetic Acid
10.3.1.6. Quaternary Ammonium Compounds
10.3.1.7. Others
10.3.2. Detergents
10.4. North America Medical Device Cleaning Market Value and Forecast, by Process
10.4.1. Manual Cleaning
10.4.2. Automatic Cleaning
10.5. North America Medical Device Cleaning Market Value and Forecast, by Application
10.5.1. Surgical Instruments
10.5.2. Ultrasound Probes
10.5.3. Endoscopes
10.5.4. Others
10.6. North America Medical Device Cleaning Market Forecast, by Country
10.6.1. U.S.
10.6.2. Canada
10.7. North America Medical Device Cleaning Market Attractiveness Analysis
10.7.1. By Product
10.7.2. By Process
10.7.3. By Application
10.7.4. By Country
11. Europe Medical Device Cleaning Market Analysis
11.1. Europe Medical Device Cleaning Market
11.2. Europe Medical Device Cleaning Market Overview
11.3. Europe Medical Device Cleaning Market Value and Forecast, by Product
11.3.1. Chemicals
11.3.1.1. Aldehydes
11.3.1.2. Hydrogen Peroxide
11.3.1.3. Alcohol
11.3.1.4. Chlorine based Disinfectant
11.3.1.5. Peracetic Acid
11.3.1.6. Quaternary Ammonium Compounds
11.3.1.7. Others
11.3.2. Detergents
11.4. Europe Medical Device Cleaning Market Value and Forecast, by Process
11.4.1. Manual Cleaning
11.4.2. Automatic Cleaning
11.5. Europe Medical Device Cleaning Market Value and Forecast, by Application
11.5.1. Surgical Instruments
11.5.2. Ultrasound Probes
11.5.3. Endoscopes
11.5.4. Others
11.6. Europe Medical Device Cleaning Market Forecast, by Country/Sub-region
11.6.1. U.K.
11.6.2. France
11.6.3. Germany
11.6.4. Spain
11.6.5. Italy
11.6.6. Rest of Europe
11.7. Europe Medical Device Cleaning Market Attractiveness Analysis
11.7.1. By Product
11.7.2. By Process
11.7.3. By Application
11.7.4. By Country/Sub-region
12. Asia Pacific Medical Device Cleaning Market Analysis
12.1. Asia Pacific Medical Device Cleaning Market
12.2. Asia Pacific Medical Device Cleaning Market Overview
12.3. Asia Pacific Medical Device Cleaning Market Value and Forecast, by Product
12.3.1. Chemicals
12.3.1.1. Aldehydes
12.3.1.2. Hydrogen Peroxide
12.3.1.3. Alcohol
12.3.1.4. Chlorine based Disinfectant
12.3.1.5. Peracetic Acid
12.3.1.6. Quaternary Ammonium Compounds
12.3.1.7. Others
12.3.2. Detergents
12.4. Asia Pacific Medical Device Cleaning Market Value and Forecast, by Process
12.4.1. Manual Cleaning
12.4.2. Automatic Cleaning
12.5. Asia Pacific Medical Device Cleaning Market Value and Forecast, by Application
12.5.1. Surgical Instruments
12.5.2. Ultrasound Probes
12.5.3. Endoscopes
12.5.4. Others
12.6. Asia Pacific Medical Device Cleaning Market Forecast, by Country/Sub-region
12.6.1. China
12.6.2. India
12.6.3. Japan
12.6.4. Australia & New Zealand
12.6.5. Rest of Asia Pacific
12.7. Asia Pacific Medical Device Cleaning Market Attractiveness Analysis
12.7.1. By Product
12.7.2. By Process
12.7.3. By Application
12.7.4. By Country/Sub-region
13. Latin America Medical Device Cleaning Market Analysis
13.1. Latin America Medical Device Cleaning Market
13.2. Latin America Medical Device Cleaning Market Overview
13.3. Latin America Medical Device Cleaning Market Value and Forecast, by Product
13.3.1. Chemicals
13.3.1.1. Aldehydes
13.3.1.2. Hydrogen Peroxide
13.3.1.3. Alcohol
13.3.1.4. Chlorine based Disinfectant
13.3.1.5. Peracetic Acid
13.3.1.6. Quaternary Ammonium Compounds
13.3.1.7. Others
13.3.2. Detergents
13.4. Latin America Medical Device Cleaning Market Value and Forecast, by Process
13.4.1. Manual Cleaning
13.4.2. Automatic Cleaning
13.5. Latin America Medical Device Cleaning Market Value and Forecast, by Application
13.5.1. Surgical Instruments
13.5.2. Ultrasound Probes
13.5.3. Endoscopes
13.5.4. Others
13.6. Latin America Medical Device Cleaning Market Forecast, by Country/Sub-region
13.6.1. Brazil
13.6.2. Mexico
13.6.3. Rest of Latin America
13.7. Latin America Medical Device Cleaning Market Attractiveness Analysis
13.7.1. By Product
13.7.2. By Process
13.7.3. By Application
13.7.4. By Country/Sub-region
14. Middle East & Africa Medical Device Cleaning Market Analysis
14.1. Middle East & Africa Medical Device Cleaning Market
14.2. Middle East & Africa Medical Device Cleaning Market Overview
14.3. Middle East & Africa Medical Device Cleaning Market Value and Forecast, by Product
14.3.1. Chemicals
14.3.1.1. Aldehydes
14.3.1.2. Hydrogen Peroxide
14.3.1.3. Alcohol
14.3.1.4. Chlorine based Disinfectant
14.3.1.5. Peracetic Acid
14.3.1.6. Quaternary Ammonium Compounds
14.3.1.7. Others
14.3.2. Detergents
14.4. Middle East & Africa Medical Device Cleaning Market Value and Forecast, by Process
14.4.1. Manual Cleaning
14.4.2. Automatic Cleaning
14.5. Middle East & Africa Medical Device Cleaning Market Value and Forecast, by Application
14.5.1. Surgical Instruments
14.5.2. Ultrasound Probes
14.5.3. Endoscopes
14.5.4. Others
14.6. Middle East & Africa Medical Device Cleaning Market Forecast, by Country/Sub-region
14.6.1. GCC countries
14.6.2. South Africa
14.6.3. Rest of Middle East & Africa
14.7. Middle East & Africa Medical Device Cleaning Market Attractiveness Analysis
14.7.1. By Product
14.7.2. By Process
14.7.3. By Application
14.7.4. By Country/Sub-region
15. Competitive Landscape
15.1. Company Profiles
15.1.1. 3M
15.1.1.1. Overview (HQ, Employee Strength, Business Segments)
15.1.1.2. Financials
15.1.1.3. Recent Developments
15.1.1.4. Strategy
15.1.2. Getinge AB
15.1.2.1. Overview (HQ, Employee Strength, Business Segments)
15.1.2.2. Financials
15.1.2.3. Recent Developments
15.1.2.4. Strategy
15.1.3. Cantel Medical Corp
15.1.3.1. Overview (HQ, Employee Strength, Business Segments)
15.1.3.2. Financials
15.1.3.3. Recent Developments
15.1.3.4. Strategy
15.1.4. STERIS plc.
15.1.4.1. Overview (HQ, Employee Strength, Business Segments)
15.1.4.2. Financials
15.1.4.3. Recent Developments
15.1.4.4. Strategy
15.1.5. Fortive Corporation
15.1.5.1. Overview (HQ, Employee Strength, Business Segments)
15.1.5.2. Financials
15.1.5.3. Recent Developments
15.1.5.4. Strategy
15.1.6. Oro Clean Chemie AG
15.1.6.1. Overview (HQ, Employee Strength, Business Segments)
15.1.6.2. Recent Developments
15.1.6.3. Strategy
15.1.7. Ruhof Corporation
15.1.7.1. Overview (HQ, Employee Strength, Business Segments)
15.1.7.2. Recent Developments
15.1.7.3. Strategy
15.1.8. Ecolab Inc
15.1.8.1. Overview (HQ, Employee Strength, Business Segments)
15.1.8.2. Financials
15.1.8.3. Recent Developments
15.1.8.4. Strategy
15.1.9. Dr. Weigert
15.1.9.1. Overview (HQ, Employee Strength, Business Segments)
15.1.9.2. Recent Developments
15.1.9.3. Strategy
List of Tables
TABLE 1 Global Medical Devices Cleaning Market Value (US$ Mn) Forecast, by Product, 2017–2027
TABLE 2 Global Medical Devices Cleaning Market Value (US$ Mn) Forecast, by Chemicals, 2017–2027
TABLE 3 Global Medical Devices Cleaning Market Value (US$ Mn) Forecast, by Process, 2017–2027
TABLE 4 Global Medical Devices Cleaning Market Value (US$ Mn) Forecast, by Application, 2017–2027
TABLE 5 Global Medical Devices Cleaning Market Value (US$ Mn) Forecast, by Region, 2017–2027
TABLE 6 North America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Country, 2017–2027
TABLE 7 North America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Product, 2017–2027
TABLE 8 North America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Chemicals, 2017–2027
TABLE 9 North America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Process, 2017–2027
TABLE 10 North America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Application, 2017–2027
TABLE 11 Europe Medical Device Cleaning Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
TABLE 12 Europe Medical Device Cleaning Market Value (US$ Mn) Forecast, by Product, 2017–2027
TABLE 13 Europe Medical Device Cleaning Market Value (US$ Mn) Forecast, by Chemicals, 2017–2027
TABLE 14 Europe Medical Device Cleaning Market Value (US$ Mn) Forecast, by Process, 2017–2027
TABLE 15 Europe Medical Device Cleaning Market Value (US$ Mn) Forecast, by Application, 2017–2027
TABLE 16 Asia Pacific Medical Device Cleaning Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
TABLE 17 Asia Pacific Medical Device Cleaning Market Value (US$ Mn) Forecast, by Product, 2017–2027
TABLE 18 Asia Pacific Medical Device Cleaning Market Value (US$ Mn) Forecast, by Chemicals, 2017–2027
TABLE 19 Asia Pacific Medical Device Cleaning Market Value (US$ Mn) Forecast, by Process, 2017–2027
TABLE 20 Asia Pacific Medical Device Cleaning Market Value (US$ Mn) Forecast, by Application, 2017–2027
TABLE 21 Latin America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
TABLE 22 Latin America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Product, 2017–2027
TABLE 23 Latin America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Chemicals, 2017–2027
TABLE 24 Latin America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Process, 2017–2027
TABLE 25 Latin America Medical Device Cleaning Market Value (US$ Mn) Forecast, by Application, 2017–2027
TABLE 26 Middle East & Africa Medical Device Cleaning Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
TABLE 27 Middle East & Africa Medical Device Cleaning Market Value (US$ Mn) Forecast, by Product, 2017–2027
TABLE 28 Middle East & Africa Medical Device Cleaning Market Value (US$ Mn) Forecast, by Chemicals, 2017–2027
TABLE 29 Middle East & Africa Medical Device Cleaning Market Value (US$ Mn) Forecast, by Process, 2017–2027
TABLE 30 Middle East & Africa Medical Device Cleaning Market Value (US$ Mn) Forecast, by Application, 2017–2027
List of Figures
Figure 1: Global Medical Devices Cleaning Market Value (US$ Mn) Forecast, 2019–2027
Figure 2: Global Medical Devices Cleaning Market Value Share, Process, 2018
Figure 3: Global Medical Devices Cleaning Market Value Share, Application, 2018
Figure 4: Global Medical Devices Cleaning Market Value Share, by Product, 2018
Figure 5: Global Medical Devices Cleaning Market Value Share, by Region, 2018
Figure 6: Global Medical Devices Cleaning Market Value Share (%), by Product, 2018 and 2027
Figure 7: Global Medical Devices Cleaning Market Attractiveness, by Product, 2019–2027
Figure 8: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Detergents, 2017–2027
Figure 9: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Chemicals, 2017–2027
Figure 10: Global Medical Devices Cleaning Market Value Share (%), by Chemicals, 2018 and 2027
Figure 11: Global Medical Devices Cleaning Market Value Share (%), by Process, 2018 and 2027
Figure 12: Global Medical Devices Cleaning Market Attractiveness, by Process, 2019–2027
Figure 13: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Manual Cleaning, 2017–2027
Figure 14: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Automatic Cleaning,
Figure 15: Global Medical Devices Cleaning Market Value Share Analysis, by Application, 2018 and 2027
Figure 16: Global Medical Devices Cleaning Market Attractiveness, by Application, 2019–2027
Figure 17: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Surgical Instruments, 2017–2027
Figure 18: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Ultrasound Probes, 2017–2027
Figure 19: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Endoscopes, 2017–2027
Figure 20: Global Medical Devices Cleaning Market Value (US$ Mn) and Y-o-Y Growth, by Others, 2017–2027
Figure 21: Global Medical Devices Cleaning Market Value Share Analysis, by Region, 2018 and 2027
Figure 22: Global Medical Devices Cleaning Market Attractiveness Analysis, by Region, 2019–2027
Figure 23: North America Medical Device Cleaning Market Value (US$ Mn) Forecast, 2017–2027
Figure 24: North America Medical Device Cleaning Market Value Share Analysis, by Country, 2018 and 2027
Figure 25: North America Medical Device Cleaning Market Attractiveness Analysis, by Country, 2019-2027
Figure 26: North America Medical Device Cleaning Market
Figure 27: North America Medical Device Cleaning Market
Figure 28: North America Medical Device Cleaning Market Value Share Analysis, by Process, 2018 and 2027
Figure 29: North America Medical Device Cleaning Market Attractiveness Analysis, by Process, 2019–2027
Figure 30: North America Medical Device Cleaning Market Value Share Analysis, by Application, 2018 and 2027
Figure 31: North America Medical Device Cleaning Market Attractiveness Analysis, by Application, 2019–2027
Figure 32: Europe Medical Device Cleaning Market Value (US$ Mn) Forecast, 2017–2027
Figure 33: Europe Medical Device Cleaning Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 34: Europe Medical Device Cleaning Market Attractiveness Analysis, by Country/Sub-region, 2019–2027
Figure 35: Europe Medical Device Cleaning Market Value Share Analysis, by Product, 2018 and 2027
Figure 36: Europe Medical Device Cleaning Market Attractiveness Analysis, by Product, 2019–2027
Figure 37: Europe Medical Device Cleaning Market Value Share Analysis, by Process, 2018 and 2027
Figure 38: Europe Medical Device Cleaning Market Attractiveness Analysis, by Process, 2019–2027
Figure 39: Europe Medical Device Cleaning Market Value Share Analysis, by Application, 2018 and 2027
Figure 40: Europe Medical Device Cleaning Market Attractiveness Analysis, by Application, 2019–2027
Figure 41: Asia Pacific Medical Device Cleaning Market Value (US$ Mn) Forecast, 2017–2027
Figure 42: Asia Pacific Medical Device Cleaning Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 43: Asia Pacific Medical Device Cleaning Market Attractiveness Analysis, by Country/Sub-region, 2019–2027
Figure 44: Asia Pacific Medical Device Cleaning Market Value Share Analysis, by Product, 2018 and 2027
Figure 45: Asia Pacific Medical Device Cleaning Market Attractiveness Analysis, by Product, 2019–2027
Figure 46: Asia Pacific Medical Device Cleaning Market Value Share Analysis, by Process, 2018 and 2027
Figure 47: Asia Pacific Medical Device Cleaning Market Attractiveness Analysis, by Process, 2019–2027
Figure 48: Asia Pacific Medical Device Cleaning Market Value Share Analysis, by Application, 2018 and 2027
Figure 49: Asia Pacific Medical Device Cleaning Market Attractiveness Analysis, by Application, 2019–2027
Figure 50: Latin America Medical Device Cleaning Market Value (US$ Mn) Forecast, 2017–2027
Figure 51: Latin America Medical Device Cleaning Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 52: Latin America Medical Device Cleaning Market Attractiveness Analysis, by Country/Sub-region, 2019–2027
Figure 53: Latin America Medical Device Cleaning Market Value Share Analysis, by Product, 2018 and 2027
Figure 54: Latin America Medical Device Cleaning Market Attractiveness Analysis, by Product, 2019–2027
Figure 55: Latin America Medical Device Cleaning Market Value Share Analysis, by Process, 2018 and 2027
Figure 56: Latin America Medical Device Cleaning Market Attractiveness Analysis, by Process, 2019–2027
Figure 57: Latin America Medical Device Cleaning Market Value Share Analysis, by Application, 2018 and 2027
Figure 58: Latin America Medical Device Cleaning Market Attractiveness Analysis, by Application, 2019–2027
Figure 59: Middle East & Africa Medical Device Cleaning Market Value (US$ Mn) Forecast, 2017–2027
Figure 60: Middle East & Africa Medical Device Cleaning Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 61: Middle East & Africa Medical Device Cleaning Market Attractiveness Analysis, by Country/Sub-region, 2019–2027
Figure 62: Middle East & Africa Medical Device Cleaning Market Value Share Analysis, by Product, 2018 and 2027
Figure 63: Middle East & Africa Medical Device Cleaning Market Attractiveness Analysis, by Product, 2019–2027
Figure 64: Middle East & Africa Medical Device Cleaning Market Value Share Analysis, by Process, 2018 and 2027
Figure 65: Middle East & Africa Medical Device Cleaning Market Attractiveness Analysis, by Process, 2019–2027
Figure 66: Middle East & Africa Medical Device Cleaning Market Value Share Analysis, by Application, 2018 and 2027
Figure 67: Middle East & Africa Medical Device Cleaning Market Attractiveness Analysis, by Application, 2019–2027