Reports
Reports
The severity of the septic process and the extent of organ dysfunction in patients require immediate admission to an ICU in hospital. To avoid extreme fatalities and admission to ICUs, healthcare companies in the sepsis therapeutics market are developing efficacious fourth- and fifth-generation drugs.
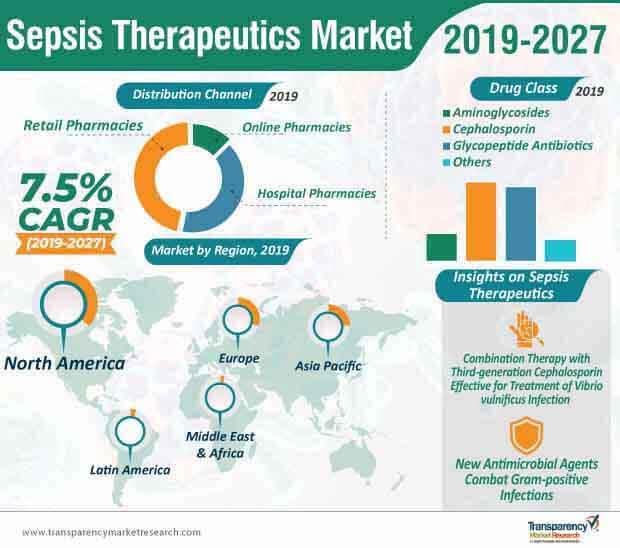
Revenue of cephalosporin drugs is projected to be the highest in 2027, in the sepsis therapeutics market, with an estimated value of ~US$ 2.2 billion. Thus, to gain positive outcome via clinical trials, manufacturers are collaborating with researchers & scientists to develop effective cephalosporin drugs. As such, healthcare institutions are approaching the combination therapy with third-generation cephalosporin and a tetracycline analogue to treat vibrio vulnificus infection in patients. This therapy has been gaining prominence in the sepsis therapeutics market, since the combination works as an 'in vitro' bactericidal activity against vibrio vulnificus.
Healthcare professionals are increasingly adopting antimicrobial therapy to minimize the damage caused by infections to the gut microbiome. As such, healthcare providers in the sepsis therapeutics market are delivering the right balance between the utilization of antimicrobials and the clinical need for treating specific signs and symptoms in patients.
Healthcare companies in the sepsis therapeutics market are increasing research on immunometabolism, as insights on immune regulation are helping manufacturers develop efficacious drugs. Since the early detection and treatment of sepsis is critical, researchers are studying the body’s natural and innate response to various classes of drugs by studying immunometabolism. To gain further insights, healthcare companies are running clinical trials involving antibiotics, fluids, and dialysis, to ensure blood flow to the affected organs. They are also devising methods of surgical therapies to remove damaged tissues.
As such, healthcare companies in the sepsis therapeutics market are trying to identify new therapeutic targets that could potentially reduce inflammatory response in patients. They are increasing research via animal testing by introducing lipopolysaccharide (LPS) in mice to see if the molecule can suppress the severity of inflammatory response.
Antimicrobials have a rapid and long-lasting effect on septic infections. However, antimicrobial resistance to gram-positive infections (GPIs) is emerging as a major public health threat to patients, worldwide. The growing prevalence of multidrug-resistant bacteria in patients has led to increased morbidity and mortality rates. This has created the issue of escalated treatment costs and prolonged hospitalization stay for patients. This is why, healthcare companies in the sepsis therapeutics market are developing new antimicrobial agents to reduce the resistance of drugs against gram-positive infections.
Healthcare companies are expected to develop a robust healthcare value and supply chain in the Asia Pacific region, as it is predicted for fast growth in the sepsis therapeutics market. For instance, stakeholders in the sepsis therapeutics market have observed the growing prevalence of methicillin resistant staphylococcus aureus bacteria in Indian patients that has led to the heavy consumption of antibiotics. Thus, stakeholders in the sepsis therapeutics market are recommending judicious prescription of antimicrobial agents, implementation of antibiotic stewardship programs, and conformation to suitable antibiotic policies developed by health commissions to reduce the problem of drug resistance in patients.

Analysts’ View of Sepsis Therapeutics Market
The sepsis therapeutics market is predicted to exponentially grow from a value of ~US$ 3.2 billion that was accounted in 2018, to a value of ~US$ 6 billion by 2027. Healthcare providers are practicing early therapeutic plasma exchange in cases of septic shock in patients. This method is considered medically safe, and effective for septic shock patients. They are also introducing high doses of vasopressors for rapid hemodynamic improvement and favorable changes in the cytokine profile for patients observed with septic shock. However, in the case of GPIs, adverse effects related to safety and tolerability towards drugs is an ongoing challenge for healthcare companies. To overcome this challenge, manufacturers should introduce new antimicrobial agents in efficacious fourth- and fifth-generation drugs.
Sepsis Therapeutics Market: Overview
Novel Pipeline Drugs and Rise in Incidence Rate of Sepsis: A Key Driver
Cephalosporin Drug Class Prominent
Intravenous Administration Popular
North America to Dominate Global Sepsis Therapeutics Market
Global Sepsis Therapeutics Market: Competitive Landscape
Global Sepsis Therapeutics Market: Key Developments
Major manufacturers in the global sepsis therapeutics market are adopting research collaboration, purchase and distribution agreement, and new product development strategies to strengthen their product portfolios and geographic presence in the international market. Key developments in the global sepsis therapeutics market are:
In the global sepsis therapeutics market report, we have discussed individual strategies, followed by company profiles of the manufacturers of sepsis drugs. The ‘competitive landscape’ section is included in the global sepsis therapeutics market report to provide readers with a dashboard view of the key players operating in the global sepsis therapeutics market.
Sepsis therapeutics market to reach a value of ~US$ 6 billion by 2027
Sepsis therapeutics market is expected to expand at a CAGR of 7.5% from 2019 to 2027
Sepsis therapeutics market is driven by increase in the number of products in the pipeline and rise in research & development expenditures
The cephalosporin segment dominated the global sepsis therapeutics market, and is expected to account for a major market share during the forecast period
Key players in the global sepsis therapeutics market include INOTREM S.A., Mylan N.V., Asahi Kasei Corporation Pfizer, Inc., GlaxoSmithKline plc
1. Preface
1.1. Market Definition and Scope
1.2. Market Segmentation
1.3. Key Research Objectives
1.4. Research Highlights
2. Assumptions and Research Methodology
3. Executive Summary: Global Sepsis Therapeutics Market
4. Market Overview
4.1. Introduction
4.1.1. Drug Class Definition
4.1.2. Industry Evolution / Developments
4.2. Overview
4.3. Market Dynamics
4.3.1. Drivers
4.3.2. Restraints
4.3.3. Opportunities
4.4. Global Sepsis Therapeutics Market Analysis and Forecasts, 2017–2027
5. Market Outlook
5.1. Regulatory Scenario
5.2. Pipeline Analysis
6. Global Sepsis Therapeutics Market Analysis and Forecasts, by Drug Class
6.1. Introduction & Definition
6.2. Key Findings / Developments
6.3. Market Value Forecast, by Drug Class, 2017–2027
6.3.1. Aminoglycosides
6.3.2. Cephalosporin
6.3.3. Glycopeptide Antibiotics
6.3.4. Others
6.4. Market Attractiveness, by Drug Class
7. Global Sepsis Therapeutics Market Analysis and Forecasts, by Route of Administration
7.1. Introduction & Definition
7.2. Key Findings / Developments
7.3. Market Value Forecast, by Route of Administration, 2017–2027
7.3.1. Intravenous
7.3.2. Oral
7.4. Market Attractiveness, by Route of Administration
8. Global Sepsis Therapeutics Market Analysis and Forecasts, by Distribution Channel
8.1. Introduction & Definition
8.2. Key Findings / Developments
8.3. Market Value Forecast, by Distribution Channel, 2017–2027
8.3.1. Hospital Pharmacies
8.3.2. Retail Pharmacies
8.3.3. Online Pharmacies
8.4. Market Attractiveness, by Distribution Channel
9. Global Sepsis Therapeutics Market Analysis and Forecasts, by Region
9.1. Key Findings
9.2. Market Value Forecast, by Region
9.2.1. North America
9.2.2. Europe
9.2.3. Asia Pacific
9.2.4. Latin America
9.2.5. Middle East & Africa
9.3. Market Attractiveness, by Region
10. North America Sepsis Therapeutics Market Analysis and Forecast
10.1. Introduction
10.1.1. Key Findings
10.2. Market Value Forecast, by Drug Class, 2017–2027
10.2.1. Aminoglycosides
10.2.2. Cephalosporin
10.2.3. Glycopeptide Antibiotics
10.2.4. Others
10.3. Market Value Forecast, by Route of Administration, 2017–2027
10.3.1. Intravenous
10.3.2. Oral
10.4. Market Value Forecast, by Distribution Channel, 2017–2027
10.4.1. Hospital Pharmacies
10.4.2. Retail Pharmacies
10.4.3. Online Pharmacies
10.5. Market Value Forecast, by Country, 2017–2027
10.5.1. U.S.
10.5.2. Canada
10.6. Market Attractiveness Analysis
10.6.1. By Drug Class
10.6.2. By Route of Administration
10.6.3. By Distribution Channel
10.6.4. By Country
11. Europe Sepsis Therapeutics Market Analysis and Forecast
11.1. Introduction
11.1.1. Key Findings
11.2. Market Value Forecast, by Drug Class, 2017–2027
11.2.1. Aminoglycosides
11.2.2. Cephalosporin
11.2.3. Glycopeptide Antibiotics
11.2.4. Others
11.3. Market Value Forecast, by Route of Administration, 2017–2027
11.3.1. Intravenous
11.3.2. Oral
11.4. Market Value Forecast, by Distribution Channel, 2017–2027
11.4.1. Hospital Pharmacies
11.4.2. Retail Pharmacies
11.4.3. Online Pharmacies
11.5. Market Value Forecast by Country/Sub-region, 2017–2027
11.5.1. Germany
11.5.2. U.K.
11.5.3. France
11.5.4. Spain
11.5.5. Italy
11.5.6. Rest of Europe
11.6. Market Attractiveness Analysis
11.6.1. By Drug Class
11.6.2. By Route of Administration
11.6.3. By Distribution Channel
11.6.4. By Country/Sub-region
12. Asia Pacific Sepsis Therapeutics Market Analysis and Forecast
12.1. Introduction
12.1.1. Key Findings
12.2. Market Value Forecast, by Drug Class, 2017–2027
12.2.1. Aminoglycosides
12.2.2. Cephalosporin
12.2.3. Glycopeptide Antibiotics
12.2.4. Others
12.3. Market Value Forecast, by Route of Administration, 2017–2027
12.3.1. Intravenous
12.3.2. Oral
12.4. Market Value Forecast, by Distribution Channel, 2017–2027
12.4.1. Hospital Pharmacies
12.4.2. Retail Pharmacies
12.4.3. Online Pharmacies
12.5. Market Value Forecast, by Country/Sub-region, 2017–2027
12.5.1. China
12.5.2. Japan
12.5.3. India
12.5.4. Australia & New Zealand
12.5.5. Rest of Asia Pacific
12.6. Market Attractiveness Analysis
12.6.1. By Drug Class
12.6.2. By Route of Administration
12.6.3. By Distribution Channel
12.6.4. By Country/Sub-region
13. Latin America Sepsis Therapeutics Market Analysis and Forecast
13.1. Introduction
13.1.1. Key Findings
13.2. Market Value Forecast, by Drug Class, 2017–2027
13.2.1. Aminoglycosides
13.2.2. Cephalosporin
13.2.3. Glycopeptide Antibiotics
13.2.4. Others
13.3. Market Value Forecast, by Route of Administration, 2017–2027
13.3.1. Intravenous
13.3.2. Oral
13.4. Market Value Forecast, by Distribution Channel, 2017–2027
13.4.1. Hospital Pharmacies
13.4.2. Retail Pharmacies
13.4.3. Online Pharmacies
13.5. Market Value Forecast, by Country/Sub-region, 2017–2027
13.5.1. Brazil
13.5.2. Mexico
13.5.3. Rest of Latin America
13.6. Market Attractiveness Analysis
13.6.1. By Drug Class
13.6.2. By Route of Administration
13.6.3. By Distribution Channel
13.6.4. By Country/Sub-region
14. Middle East & Africa Sepsis Therapeutics Market Analysis and Forecast
14.1. Introduction
14.1.1. Key Findings
14.2. Market Value Forecast, by Drug Class, 2017–2027
14.2.1. Aminoglycosides
14.2.2. Cephalosporin
14.2.3. Glycopeptide Antibiotics
14.2.4. Others
14.3. Market Value Forecast, by Route of Administration, 2017–2027
14.3.1. Intravenous
14.3.2. Oral
14.4. Market Value Forecast, by Distribution Channel, 2017–2027
14.4.1. Hospital Pharmacies
14.4.2. Retail Pharmacies
14.4.3. Online Pharmacies
14.5. Market Value Forecast, by Country/Sub-region, 2017–2027
14.5.1. GCC Countries
14.5.2. South Africa
14.5.3. Rest of Middle East & Africa
14.6. Market Attractiveness Analysis
14.6.1. By Drug Class
14.6.2. By Route of Administration
14.6.3. By Distribution Channel
14.6.4. By Country/Sub-region
15. Competitive Landscape
15.1. Market Player – Competition Matrix (By Tier and Size of companies)
15.2. Company Profiles
15.2.1. Mylan N.V.
15.2.1.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.1.2. Product Portfolio
15.2.1.3. SWOT Analysis
15.2.1.4. Strategic Overview
15.2.1.5. Financial Overview
15.2.2. Asahi Kasei Corporation
15.2.2.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.2.2. Product Portfolio
15.2.2.3. SWOT Analysis
15.2.2.4. Strategic Overview
15.2.2.5. Financial Overview
15.2.3. Pfizer, Inc.
15.2.3.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.3.2. Product Portfolio
15.2.3.3. SWOT Analysis
15.2.3.4. Strategic Overview
15.2.3.5. Financial Overview
15.2.4. GlaxoSmithKline plc
15.2.4.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.4.2. Product Portfolio
15.2.4.3. SWOT Analysis
15.2.4.4. Strategic Overview
15.2.4.5. Financial Overview
15.2.5. RegeneRx
15.2.5.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.5.2. Product Portfolio
15.2.5.3. SWOT Analysis
15.2.5.4. Strategic Overview
15.2.5.5. Financial Overview
15.2.6. Adrenomed AG
15.2.6.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.6.2. Product Portfolio
15.2.6.3. Strategic Overview
15.2.7. AtoxBio
15.2.7.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.8. ENDACEA, INC.
15.2.8.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.8.2. Product Portfolio
15.2.8.3. Strategic Overview
15.2.9. INOTREM S.A
15.2.9.1. Company Overview (HQ, Business Segments, Employee Strength)
15.2.9.2. Product Portfolio
15.2.9.3. Strategic Overview
List of Tables
Table 01: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Drug Class, 2017–2027
Table 02: Global Sepsis Therapeutics Drugs Market Value (US$ Mn) Forecast, by Route of Administration, 2017–2027
Table 03: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Distribution Channel, 2017–2027
Table 04: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Region, 2017–2027
Table 05: North America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Country, 2017–2027
Table 06: North America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Drug Class, 2017–2027
Table 07: North America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Route of Administration, 2017–2027
Table 08: North America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Distribution Channel, 2017–2027
Table 09: Europe Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
Table 10: Europe Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Drug Class, 2017–2027
Table 11: Europe Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Route of Administration, 2017–2027
Table 12: Europe Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Distribution Channel, 2017–2027
Table 13: Asia Pacific Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
Table 14: Asia Pacific Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Drug Class, 2017–2027
Table 15: Asia Pacific Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Route of Administration, 2017–2027
Table 16: Asia Pacific Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Distribution Channel, 2017–2027
Table 17: Latin America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
Table 18: Latin America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Drug Class, 2017–2027
Table 19: Latin America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Route of Administration, 2017–2027
Table 20: Latin America Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Distribution Channel, 2017–2027
Table 21: Middle East & Africa Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
Table 22: Middle East & Africa Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Drug Class, 2017–2027
Table 23: Middle East & Africa Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Route of Administration, 2017–2027
Table 24: Middle East & Africa Sepsis Therapeutics Market Value (US$ Mn) Forecast, by Distribution Channel, 2017–2027
List of Figures
Figure 01: Global Sepsis Therapeutics Market Value (US$ Mn) and Distribution, by Region, 2018 and 2027
Figure 02: Global Sepsis Therapeutics Market Revenue (US$ Mn), by Route of Administration, 2018
Figure 04: Global Sepsis Therapeutics Market Revenue, by Drug Class (US$ Mn), 2018
Figure 03: Global Sepsis Therapeutics Market Revenue (US$ Mn), by Distribution Channel, 2018
Figure 05: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast, 2017–2027
Figure 07: Global Sepsis Therapeutics Market Value Share, by Route of Administration, 2018
Figure 09: Global Sepsis Therapeutics Market Value Share, by Region, 2018
Figure 06: Global Sepsis Therapeutics Market Value Share, by Drug Class, 2018
Figure 08: Global Sepsis Therapeutics Market Value Share, by Distribution Channel, 2018
Figure 10: Regulatory Approval Process - U.S.
Figure 11: Regulatory Approval Process - Canada
Figure 12: Regulatory Approval Process - Europe
Figure 13: Regulatory Approval Process - Japan
Figure 14: Regulatory Approval Process - India
Figure 15: Global Sepsis Therapeutics Market Value Share Analysis, by Drug Class, 2018 and 2027
Figure 16: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast and Y-o-Y Growth (%), by Aminoglycosides, 2017–2027
Figure 17: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast and Y-o-Y Growth (%), by Cephalosporin, 2017–2027
Figure 18: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast and Y-o-Y Growth (%), by Glycopeptide Antibiotics, 2017–2027
Figure 19: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast and Y-o-Y Growth (%), by Others, 2017–2027
Figure 20: Global Sepsis Therapeutics Market Attractiveness, by Drug Class, 2019–2027
Figure 21: Global Sepsis Therapeutics Market Value Share (%), by Route of Administration, 2018 and 2027
Figure 22: Global Sepsis Therapeutics Drugs Market Value (US$ Mn) Forecast and Y-o-Y Growth (%), by Intravenous, 2017–2027
Figure 23: Global Sepsis Therapeutics Drugs Market Value (US$ Mn) Forecast and Y-o-Y Growth (%), by Oral, 2017–2027
Figure 24: Global Sepsis Therapeutics Market Attractiveness, by Route of Administration, 2019–2027
Figure 25: Global Sepsis Therapeutics Market Value Share (%), by Distribution Channel, 2018 and 2027
Figure 26: Global Sepsis Therapeutics Market Revenue (US$ Mn) Forecast and Y-o-Y Growth (%), by Hospital Pharmacies, 2017–2027
Figure 27: Global Sepsis Therapeutics Market Revenue (US$ Mn) Forecast and Y-o-Y Growth (%), by Retail Pharmacies, 2017–2027
Figure 28: Global Sepsis Therapeutics Market Value (US$ Mn) Forecast and Y-o-Y Growth (%), by Online Pharmacies, 2017–2027
Figure 29: Global Sepsis Therapeutics Market Attractiveness, by Distribution Channel, 2019–2027
Figure 30: Global Sepsis Therapeutics Market Value Share Analysis, by Region 2018 and 2027
Figure 31: Global Sepsis Therapeutics Market Attractiveness, by Region, 2019–2027
Figure 32: North America Sepsis Therapeutics Market Value (US$ Mn) Forecast and Y-o-Y Growth (%) Projection, 2017–2027
Figure 33: North America Sepsis Therapeutics Market Value Share (%) Analysis, by Country, 2018 and 2027
Figure 34: North America Sepsis Therapeutics Market Attractiveness Analysis, by Country, 2019?2027
Figure 35: North America Sepsis Therapeutics Market Value Share (%) Analysis, by Drug Class, 2018 and 2027
Figure 36: North America Sepsis Therapeutics Market Attractiveness Analysis, by Drug Class, 2019–2027
Figure 37: North America Sepsis Therapeutics Market Value Share (%) Analysis, by Route of Administration, 2018 and 2027
Figure 38: North America Sepsis Therapeutics Market Attractiveness Analysis, by Route of Administration, 2019–2027
Figure 39: North America Sepsis Therapeutics Market Value Share (%) Analysis, by Distribution Channel, 2018 and 2027
Figure 40: North America Sepsis Therapeutics Market Attractiveness Analysis, by Distribution Channel, 2019–2027
Figure 41: Europe Sepsis Therapeutics Market Value (US$ Mn) and Y-o-Y Growth (%) Forecast, 2017–2027
Figure 42: Europe Sepsis Therapeutics Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 43: Europe Sepsis Therapeutics Market Attractiveness Analysis, by Country/Sub-region, 2019?2027
Figure 44: Europe Sepsis Therapeutics Market Value Share Analysis (%), by Drug Class, 2018 and 2027
Figure 45: Europe Sepsis Therapeutics Market Attractiveness, by Drug Class, 2019–2027
Figure 46: Europe Sepsis Therapeutics Market Value Share Analysis (%), by Route of Administration, 2018 and 2027
Figure 47: Europe Sepsis Therapeutics Market Attractiveness, by Route of Administration, 2019–2027
Figure 48: Europe Sepsis Therapeutics Market Value Share Analysis (%), by Distribution Channel, 2018 and 2027
Figure 49: Europe Sepsis Therapeutics Market Attractiveness, by Distribution Channel, 2019–2027
Figure 50: Asia Pacific Sepsis Therapeutics Market Value (US$ Mn) and Y-o-Y Growth (%) Forecast, 2017–2027
Figure 51: Asia Pacific Sepsis Therapeutics Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 52: Asia Pacific Sepsis Therapeutics Market Attractiveness Analysis, by Country/Sub-region, 2019–2027
Figure 53: Asia Pacific Sepsis Therapeutics Market Value Share Analysis (%), by Drug Class, 2018 and 2027
Figure 54: Asia Pacific Sepsis Therapeutics Market Attractiveness, by Drug Class, 2019–2027
Figure 55: Asia Pacific Sepsis Therapeutics Market Value Share Analysis (%), by Route of Administration, 2018 and 2027
Figure 56: Asia Pacific Sepsis Therapeutics Market Attractiveness, by Route of Administration, 2019–2027
Figure 57: Asia Pacific Sepsis Therapeutics Market Value Share Analysis (%), by Distribution Channel, 2018 and 2027
Figure 58: Asia Pacific Sepsis Therapeutics Market Attractiveness, by Distribution Channel, 2019–2027
Figure 59: Latin America Sepsis Therapeutics Market Value (US$ Mn) and Y-o-Y Growth (%) Forecast, 2017–2027
Figure 60: Latin America Sepsis Therapeutics Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 61: Latin America Sepsis Therapeutics Market Attractiveness Analysis, by Country/Sub-region, 2019–2027
Figure 62: Latin America Sepsis Therapeutics Market Value Share Analysis (%), by Drug Class, 2018 and 2027
Figure 63: Latin America Sepsis Therapeutics Market Attractiveness, by Drug Class, 2019–2027
Figure 64: Latin America Sepsis Therapeutics Market Value Share Analysis (%), by Route of Administration, 2018 and 2027
Figure 65: Latin America Sepsis Therapeutics Market Attractiveness, by Route of Administration, 2019–2027
Figure 66: Latin America Sepsis Therapeutics Market Value Share Analysis (%), by Distribution Channel, 2018 and 2027
Figure 67: Latin America Sepsis Therapeutics Market Attractiveness, by Distribution Channel, 2019–2027
Figure 68: Middle East & Africa Sepsis Therapeutics Market Value (US$ Mn) and Y-o-Y Growth (%) Forecast, 2017–2027
Figure 69: Middle East & Africa Sepsis Therapeutics Market Value Share Analysis, by Country/Sub-region, 2018 and 2027
Figure 70: Middle East & Africa Sepsis Therapeutics Market Attractiveness Analysis, by Country/Sub-region, 2019-2027
Figure 71: Middle East & Africa Sepsis Therapeutics Market Value Share Analysis (%), by Drug Class, 2018 and 2027
Figure 72: Middle East & Africa Sepsis Therapeutics Market Attractiveness, by Drug Class, 2019–2027
Figure 73: Middle East & Africa Sepsis Therapeutics Market Value Share Analysis (%), by Route of Administration, 2018 and 2027
Figure 74: Middle East & Africa Sepsis Therapeutics Market Attractiveness, by Route of Administration, 2019–2027
Figure 75: Middle East & Africa Sepsis Therapeutics Market Value Share Analysis (%), by Distribution Channel, 2018 and 2027
Figure 76: Middle East & Africa Sepsis Therapeutics Market Attractiveness, by Distribution Channel, 2019–2027