Reports
Reports
Analyst Viewpoint
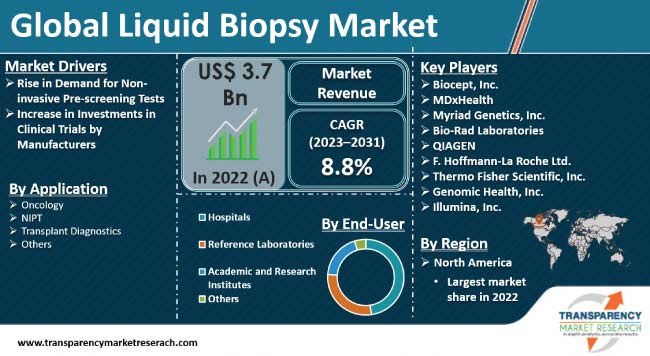
Rise in prevalence of cancer, increase in applications of liquid biopsy and growing research on enhancing efficacy of liquid biopsy over other diagnostic tests are estimated to propel the market in 2023. Furthermore, increase in demand for early cancer detection through non-invasive procedures is also likely to fuel the liquid biopsy market demand in the next few years.
Leading companies in liquid biopsy field are collaborating to establish industry-wide standards and comply with stringent regulatory requirements. Moreover, advancements in multi-omics analysis, expansion of liquid biopsy applications to other diseases, and the integration of artificial intelligence for data interpretation are expected to boost the liquid biopsy industry in the next few years.
Liquid biopsies can improve or resolve several inherent issues with current cancer treatment, enabling early cancer detection, mutation detection, tumor monitoring during treatment. They have the potential to detect cancer before tumor formation, assess multiple mutations within the tumor.
Governments are taking initiatives regarding public screening tests to reduce the further spread of chronic disorders. Non-invasive screening tests help in detection of signs of cancerous tumors inclusive of cell free DNAs, circulating biomarkers, and circulating tumor DNAs (ctDNAs) in cancer patients at recurrent or screening stages. Growing frequency of US FDA-approved advanced tests is expected to drive liquid biopsy market revenue in the next few years.
The ability of liquid biopsies to capture tumor heterogeneity and dynamics, detect MRD at an early stage, assess real-time treatment response, biomarker discovery, and non-invasive nature make liquid biopsy test an invaluable tool in formulating personalized treatment strategies. Liquid biopsy for cancer test also allows for easier accessibility, especially when obtaining tissue biopsies may be challenging or impractical. Therefore, rise in demand for non-invasive diagnostic approaches is anticipated to offer significant liquid biopsy market opportunities in the near future.
The non-invasive nature of liquid biopsies eliminates the need for traditional tissue biopsies, reducing patient discomfort and the risk of complications. This makes liquid biopsies particularly appealing for patients who may be reluctant or unable to undergo surgical procedures. Ongoing research is focused on boosting the application of liquid biopsies to population-wide cancer screening programs. The potential for early detection of cancer in asymptomatic individuals could significantly impact cancer mortality rates.
Demand for non-invasive prenatal testing is considerably high due to an increase in number of congenital and genetic abnormalities in the fetus. According to an article published by the UN group, in Arch 2022, nearly 3000 to 5000 children are born with Down syndrome annually. In May 2021, Yourgene announced the launch of IONA care – a NIPT service offering.
This solution provides an extended advantage to measure the probability that a pregnant woman is carrying fetus with autosomal aneuploidies (AA2) and sex chromosome aneuploidies (SCA1) apart from screening for trisomies 13, 18 and 21 and fetal sex determination. Thus, growing awareness about the importance of prenatal testing is expected to fuel the liquid biopsy market development in the next few years.
Currently, technology advancements in liquid biopsies are driving the frequency of therapeutic selection, cancer screening, recurrence monitoring, and drug trial optimization. Several government agencies, in collaboration with key players, are emphasizing investment in R&D to conduct clinical studies on liquid biopsy for offering precision medicine for patients.
In November 2021, BioMark Diagnostics, Inc. received funding close to US$ 135,640 from National Research Council of Canada Industrial Research Assistance Program (NRCIRAP). The basic purpose of this funding was to extend support to R&D of liquid biopsy assay to screen and detect lung cancer at early stages.
In November 2022, Thermo Fisher Scientific tabled digital PCR liquid-biopsy assays for Applied Biosystems absolute Q dPCR system and custom design tool for simplifying cancer research in clinical research and academic institutes.
Investments in liquid biopsy testing for early cancer detection have been rising for the last few years, as it provides valuable insights into the molecular characteristics of tumors, enabling the tailoring of cancer treatments based on individual genetic profiles. This personalized approach holds promise for more effective and targeted therapies and is consequently expected to bolster the global liquid biopsy market development during the forecast period.
North America accounted for the largest liquid biopsy market share in 2022 due to growing incidence of cancer and rapid advancements in screening technology in the region. As per the American Cancer Society, 1.9 million cancer cases with 609,360 fatalities were recorded in the U.S in 2022. Several organizations such as the American Society of Clinical Oncology (ASCO) are working on creating awareness campaigns regarding utilization of various types of liquid biopsy tests for early cancer detection.
According to the latest market analysis, growing awareness about prenatal screening and early cancer detection among the population is estimated to positively impact the liquid biopsy market outlook in Europe in the next few years.
Governments across Europe are taking initiatives to boost cancer screening along with various other pathology tests. In March 2022, the U.K. National health Service offered liquid biopsy tests to some cancer patients for complementing anatomic pathology testing with an effort of diagnosing 75% of all kinds of cancer at stage I or stage II by 2028. Moreover, as per the data by published by MDPI in 2022, close to 6,560 patients benefit from liquid biopsy tests every year.
Leading companies are following the latest liquid biopsy market trends and focusing on research and development initiatives, strategic collaborations, and the commercialization of advanced liquid biopsy technologies. Key players are engaged in organic as well as inorganic modes of expansion to strengthen their global presence. Biocept, Inc., MDxHealth, Myriad Genetics, Inc., Bio-Rad Laboratories, QIAGEN, F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific, Inc., Genomic Health, Inc., and Illumina, Inc. are a few prominent entities operating in the global market.
Key players in the liquid biopsy market report have been profiled based on various parameters such as company overview, business strategies, financial overview, product portfolio, and business segments.
| Attribute | Detail |
|---|---|
| Size in 2022 | US$ 3.7 Bn |
| Forecast (Value) in 2031 | US$ 7.7 Bn |
| Growth Rate (CAGR) | 8.8% |
| Forecast Period | 2023-2031 |
| Historical Data Available for | 2017-2021 |
| Quantitative Units | US$ Bn for Value |
| Market Analysis | It includes segment analysis as well as regional level analysis. Moreover, qualitative analysis includes drivers, restraints, opportunities, key trends, Porter’s Five Forces analysis, value chain analysis, and key trend analysis. |
| Competition Landscape |
|
| Format | Electronic (PDF) + Excel |
| Market Segmentation |
|
| Regions Covered |
|
| Countries Covered |
|
| Companies Profiled |
|
| Customization Scope | Available Upon Request |
| Pricing | Available Upon Request |
The global market was valued at US$ 3.7 Bn in 2022
It is projected to grow at a CAGR of 8.8% from 2023 to 2031
Rise in demand for non-invasive pre-natal tests and increase in investments in clinical trials
In terms of end-user, the hospitals segment held largest share in 2022
North America is estimated to dominate in the next few years
Biocept, Inc., MDxHealth, Myriad Genetics, Inc., Bio-Rad Laboratories, QIAGEN, F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific, Inc., Genomic Health, Inc., and Illumina, Inc.
1. Preface
1.1. Market Definition and Scope
1.2. Market Segmentation
1.3. Key Research Objectives
1.4. Research Highlights
2. Assumptions
3. Research Methodology
4. Executive Summary
5. Market Overview
5.1. Introduction
5.2. Market Dynamics
5.2.1. Drivers
5.2.2. Restraints
5.2.3. Opportunities
5.3. Key Trends Analysis
5.3.1. Demand Side Analysis
5.3.2. Supply Side Analysis
5.4. Key Market Indicators
5.5. Technological Overview Analysis
5.6. Porter’s Five Forces Analysis
5.7. Industry SWOT Analysis
5.8. Value Chain Analysis
5.9. Regulatory Framework
5.10. Global Liquid Biopsy Market Analysis and Forecast, - 2017–2031
5.10.1. Market Value Projections (US$ Mn)
6. Global Liquid Biopsy Market Analysis and Forecast, by Application
6.1. Liquid Biopsy Market Size (US$ Mn) Forecast, by Application, 2017–2031
6.1.1. Oncology
6.1.2. NIPT
6.1.3. Transplant Diagnostics
6.1.4. Others
6.2. Incremental Opportunity, by Application
7. Global Liquid Biopsy Market Analysis and Forecast, by Circulating Biomarker
7.1. Liquid Biopsy Market Size (US$ Mn) Forecast, by Circulating Biomarker, 2017–2031
7.1.1. Circulating Tumor Cells (CTC)
7.1.2. Free Nucleic Acid
7.1.3. Exracellular Vesicles/Exosomes
7.2. Incremental Opportunity, by Circulating Biomarker
8. Global Liquid Biopsy Market Analysis and Forecast, by End-user
8.1. Liquid Biopsy Market Size (US$ Mn) Forecast, End-user, 2017–2031
8.1.1. Hospitals
8.1.2. Reference Laboratories
8.1.3. Academic and Research Institutes
8.1.4. Others
8.2. Incremental Opportunity, by End-user
9. Global Liquid Biopsy Market Analysis and Forecast, by Region
9.1. Liquid Biopsy Market Size (US$ Mn) Forecast, by Region, 2017–2031
9.1.1. North America
9.1.2. Europe
9.1.3. Asia Pacific
9.1.4. Middle East & Africa
9.1.5. South America
9.2. Incremental Opportunity, by Region
10. North America Liquid Biopsy Market Analysis and Forecast
10.1. Regional Snapshot
10.2. Brand Analysis
10.3. Price Trend Analysis
10.3.1. Weighted Average Price
10.4. Key Trends Analysis
10.4.1. Demand Side
10.4.2. Supplier Side
10.5. Liquid Biopsy Market Size (US$ Mn) Forecast, by Application, 2017–2031
10.5.1. Oncology
10.5.2. NIPT
10.5.3. Transplant Diagnostics
10.5.4. Others
10.6. Liquid Biopsy Market Size (US$ Mn) Forecast, by Circulating Biomarker, 2017–2031
10.6.1. Circulating Tumor Cells (CTC)
10.6.2. Free Nucleic Acid
10.6.3. Exracellular Vesicles/Exosomes
10.7. Liquid Biopsy Market Size (US$ Mn) Forecast, End-user - 2017–2031
10.7.1. Hospitals
10.7.2. Reference Laboratories
10.7.3. Academic and Research Institutes
10.7.4. Others
10.8. Liquid Biopsy Market Size (US$ Mn) Forecast, by Country - 2017–2031
10.8.1. U.S.
10.8.2. Canada
10.8.3. Rest of North America
10.9. Incremental Opportunity Analysis
11. Europe Liquid Biopsy Market Analysis and Forecast
11.1. Regional Snapshot
11.2. Brand Analysis
11.3. Price Trend Analysis
11.3.1. Weighted Average Price
11.4. Key Trends Analysis
11.4.1. Demand Side
11.4.2. Supplier Side
11.5. Liquid Biopsy Market Size (US$ Mn) Forecast, by Application, 2017–2031
11.5.1. Oncology
11.5.2. NIPT
11.5.3. Transplant Diagnostics
11.5.4. Others
11.6. Liquid Biopsy Market Size (US$ Mn) Forecast, by Circulating Biomarker, 2017–2031
11.6.1. Circulating Tumor Cells (CTC)
11.6.2. Free Nucleic Acid
11.6.3. Exracellular Vesicles/Exosomes
11.7. Liquid Biopsy Market Size (US$ Mn) Forecast, End-user, 2017–2031
11.7.1. Hospitals
11.7.2. Reference Laboratories
11.7.3. Academic and Research Institutes
11.7.4. Others
11.8. Liquid Biopsy Market Size (US$ Mn) Forecast, by Country, 2017–2031
11.8.1. U.K.
11.8.2. Germany
11.8.3. France
11.8.4. Rest of Europe
11.9. Incremental Opportunity Analysis
12. Asia Pacific Liquid Biopsy Market Analysis and Forecast
12.1. Regional Snapshot
12.2. Brand Analysis
12.3. Price Trend Analysis
12.3.1. Weighted Average Price
12.4. Key Trends Analysis
12.4.1. Demand Side
12.4.2. Supplier Side
12.5. Liquid Biopsy Market Size (US$ Mn) Forecast, by Application, 2017–2031
12.5.1. Oncology
12.5.2. NIPT
12.5.3. Transplant Diagnostics
12.5.4. Others
12.6. Liquid Biopsy Market Size (US$ Mn) Forecast, by Circulating Biomarker, 2017–2031
12.6.1. Circulating Tumor Cells (CTC)
12.6.2. Free Nucleic Acid
12.6.3. Exracellular Vesicles/Exosomes
12.7. Liquid Biopsy Market Size (US$ Mn) Forecast, End-user, 2017–2031
12.7.1. Hospitals
12.7.2. Reference Laboratories
12.7.3. Academic and Research Institutes
12.7.4. Others
12.8. Liquid Biopsy Market Size (US$ Mn) Forecast, by Country, 2017–2031
12.8.1. India
12.8.2. China
12.8.3. Japan
12.8.4. Rest of Asia Pacific
12.9. Incremental Opportunity Analysis
13. Middle East & South Africa Liquid Biopsy Market Analysis and Forecast
13.1. Regional Snapshot
13.2. Brand Analysis
13.3. Price Trend Analysis
13.3.1. Weighted Average Price
13.4. Key Trends Analysis
13.4.1. Demand Side
13.4.2. Supplier Side
13.5. Liquid Biopsy Market Size (US$ Mn) Forecast, by Application, 2017–2031
13.5.1. Oncology
13.5.2. NIPT
13.5.3. Transplant Diagnostics
13.5.4. Others
13.6. Liquid Biopsy Market Size (US$ Mn) Forecast, by Circulating Biomarker, 2017–2031
13.6.1. Circulating Tumor Cells (CTC)
13.6.2. Free Nucleic Acid
13.6.3. Exracellular Vesicles/Exosomes
13.7. Liquid Biopsy Market Size (US$ Mn) Forecast, End-user, 2017–2031
13.7.1. Hospitals
13.7.2. Reference Laboratories
13.7.3. Academic and Research Institutes
13.7.4. Others
13.8. Liquid Biopsy Market Size (US$ Mn) Forecast, by Country, 2017–2031
13.8.1. GCC
13.8.2. Rest of Middle East & Africa
13.9. Incremental Opportunity Analysis
14. South America Liquid Biopsy Market Analysis and Forecast
14.1. Regional Snapshot
14.2. Brand Analysis
14.3. Price Trend Analysis
14.3.1. Weighted Average Price
14.4. Key Trends Analysis
14.4.1. Demand Side
14.4.2. Supplier Side
14.5. Liquid Biopsy Market Size (US$ Mn) Forecast, by Application, 2017–2031
14.5.1. Oncology
14.5.2. NIPT
14.5.3. Transplant Diagnostics
14.5.4. Others
14.6. Liquid Biopsy Market Size (US$ Mn) Forecast, by Circulating Biomarker, 2017–2031
14.6.1. Circulating Tumor Cells (CTC)
14.6.2. Free Nucleic Acid
14.6.3. Exracellular Vesicles/Exosomes
14.7. Liquid Biopsy Market Size (US$ Mn) Forecast, End-user, 2017–2031
14.7.1. Hospitals
14.7.2. Reference Laboratories
14.7.3. Academic and Research Institutes
14.7.4. Others
14.8. Liquid Biopsy Market Size (US$ Mn) Forecast, by Country, 2017–2031
14.8.1. Brazil
14.8.2. Rest of South America
14.9. Incremental Opportunity Analysis
15. Competition Landscape
15.1. Market Player - Competition Dashboard
15.2. Market Share Analysis (%), by Company, (2022)
15.3. Company Profiles (Details - Company Overview, Sales Area/Geographical Presence, Revenue, Strategy & Business Overview)
15.3.1. Biocept, Inc.
15.3.1.1. Company Overview
15.3.1.2. Sales Area
15.3.1.3. Geographical Presence
15.3.1.4. Revenue
15.3.1.5. Strategy & Business Overview
15.3.2. MDxHealth
15.3.2.1. Company Overview
15.3.2.2. Sales Area
15.3.2.3. Geographical Presence
15.3.2.4. Revenue
15.3.2.5. Strategy & Business Overview
15.3.3. Myriad Genetics, Inc.
15.3.3.1. Company Overview
15.3.3.2. Sales Area
15.3.3.3. Geographical Presence
15.3.3.4. Revenue
15.3.3.5. Strategy & Business Overview
15.3.4. Bio-Rad Laboratories
15.3.4.1. Company Overview
15.3.4.2. Sales Area
15.3.4.3. Geographical Presence
15.3.4.4. Revenue
15.3.4.5. Strategy & Business Overview
15.3.5. QIAGEN
15.3.5.1. Company Overview
15.3.5.2. Sales Area
15.3.5.3. Geographical Presence
15.3.5.4. Revenue
15.3.5.5. Strategy & Business Overview
15.3.6. F. Hoffmann-La Roche Ltd.
15.3.6.1. Company Overview
15.3.6.2. Sales Area
15.3.6.3. Geographical Presence
15.3.6.4. Revenue
15.3.6.5. Strategy & Business Overview
15.3.7. Thermo Fisher Scientific, Inc.
15.3.7.1. Company Overview
15.3.7.2. Sales Area
15.3.7.3. Geographical Presence
15.3.7.4. Revenue
15.3.7.5. Strategy & Business Overview
15.3.8. Genomic Health, Inc.
15.3.8.1. Company Overview
15.3.8.2. Sales Area
15.3.8.3. Geographical Presence
15.3.8.4. Revenue
15.3.8.5. Strategy & Business Overview
15.3.9. Illumina, Inc.
15.3.9.1. Company Overview
15.3.9.2. Sales Area
15.3.9.3. Geographical Presence
15.3.9.4. Revenue
15.3.9.5. Strategy & Business Overview
15.3.10. Other Key Players
15.3.10.1. Company Overview
15.3.10.2. Sales Area
15.3.10.3. Geographical Presence
15.3.10.4. Revenue
15.3.10.5. Strategy & Business Overview
16. Go to Market Strategy
17. Identification of Potential Market Spaces
18. Prevailing Market Risks
19. Understanding Buying Process of Customers
20. Preferred Sales & Marketing Strategy
List of Tables
Table 1: Global Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Table 2: Global Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Table 3: Global Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Table 4: Global Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Table 5: North America Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Table 6: North America Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Table 7: North America Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Table 8: North America Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Table 9: Europe Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Table 10: Europe Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Table 11: Europe Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Table 12: Europe Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Table 13: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Table 14: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Table 15: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Table 16: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Table 17: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Table 18: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Table 19: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Table 20: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Table 21: South America Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Table 22: South America Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Table 23: South America Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Table 24: South America Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
List of Figures
Figure 1: Global Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Figure 2: Global Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Application, 2023-2031
Figure 3: Global Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Figure 4: Global Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Circulating Biomarker, 2023-2031
Figure 5: Global Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Figure 6: Global Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by End-user, 2023-2031
Figure 7: Global Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Figure 8: Global Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Region, 2023-2031
Figure 9: North America Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Figure 10: North America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Application, 2023-2031
Figure 11: North America Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Figure 12: North America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Circulating Biomarker, 2023-2031
Figure 13: North America Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Figure 14: North America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by End-user, 2023-2031
Figure 15: North America Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Figure 16: North America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Region, 2023-2031
Figure 17: Europe Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Figure 18: Europe Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Application, 2023-2031
Figure 19: Europe Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Figure 20: Europe Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Circulating Biomarker, 2023-2031
Figure 21: Europe Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Figure 22: Europe Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by End-user, 2023-2031
Figure 23: Europe Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Figure 24: Europe Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Region, 2023-2031
Figure 25: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Figure 26: Asia Pacific Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Application, 2023-2031
Figure 27: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Figure 28: Asia Pacific Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Circulating Biomarker, 2023-2031
Figure 29: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Figure 30: Asia Pacific Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by End-user, 2023-2031
Figure 31: Asia Pacific Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Figure 32: Asia Pacific Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Region, 2023-2031
Figure 33: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Figure 34: Middle East & Africa Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Application, 2023-2031
Figure 35: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Figure 36: Middle East & Africa Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Circulating Biomarker, 2023-2031
Figure 37: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Figure 38: Middle East & Africa Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by End-user, 2023-2031
Figure 39: Middle East & Africa Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Figure 40: Middle East & Africa Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Region, 2023-2031
Figure 41: South America Liquid Biopsy Market Value (US$ Mn), by Application, 2017-2031
Figure 42: South America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Application, 2023-2031
Figure 43: South America Liquid Biopsy Market Value (US$ Mn), by Circulating Biomarker, 2017-2031
Figure 44: South America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Circulating Biomarker, 2023-2031
Figure 45: South America Liquid Biopsy Market Value (US$ Mn), by End-user, 2017-2031
Figure 46: South America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by End-user, 2023-2031
Figure 47: South America Liquid Biopsy Market Value (US$ Mn), by Region, 2017-2031
Figure 48: South America Liquid Biopsy Market Incremental Opportunity (US$ Mn), Forecast, by Region, 2023-2031