Reports
Reports
Analyst Viewpoint
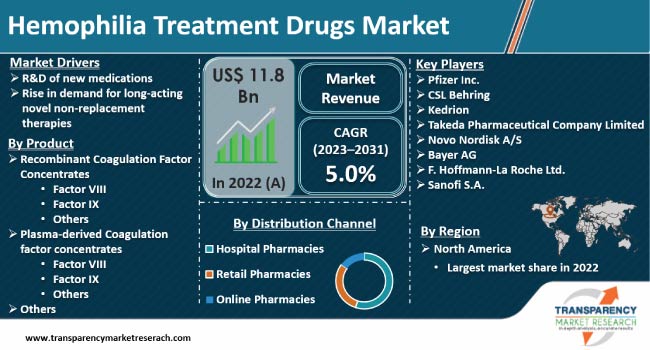
R&D of new medications and rise in demand for long-acting novel non-replacement therapies are expected to propel the hemophilia treatment drugs market size during the forecast period. Surge in diagnosis for hemophilia is also boosting demand for hemophilia treatment drugs.
R&D of personalized approaches in hemophilia treatment is likely to offer lucrative opportunities to vendors in the global hemophilia treatment drugs industry. Government bodies in various countries are funding the R&D of new drugs to reduce the burden of hemophilia. Anti-TFPI medications are gaining traction among patients as these medications aim to lower the mechanism that prevents blood from clotting excessively.
Hemophilia is an inherited bleeding illness. A person's blood does not clot effectively with this disease, which causes a variety of issues such as spontaneous bleeding. Hemophilia is most common among men. Hemophilia treatment drugs are pharmaceuticals that have been authorized by relevant regulatory bodies for the treatment of hemophilia. According to the Centers for Disease Control and Prevention (CDC), around 400 newborns in the U.S. are born with hemophilia A. According to the CDC report, about 33,000 men in the U.S. suffered from the disease. Thus, increase in cases of hemophilia is expected to spur the hemophilia treatment drugs market growth in the near future.
Research activities play a vital role in the hemophilia treatment drugs market value. Most hemophilia treatment drug manufacturers are investing heavily in these activities for the development of new formulations that are beneficial for the treatment of hemophilia.
Anti-TFPI is a novel hemophilia medication that aims to minimize bleeding by lowering (or slowing) the mechanism that prevents blood from clotting excessively. Anti-TFPI restores hemostatic equilibrium by inhibiting the function of one of the anticoagulants, TFPI. Since anticoagulants prevent clotting, interfering with how they function causes clotting to occur. Anti-TFPI is not based on the replacement of a specific clotting protein, such as factor VIII(8) or factor IX(9). As a result, this medication for hemophilia can be used to reduce bleeding episodes in patients with both hemophilia A and hemophilia B. Anti-TFPI clinical studies are now in phase 3.
RNA Interference (RNAi) treatment targets an anticoagulant, antithrombin. It is a new treatment that aims to restore hemostatic equilibrium by lowering antithrombin levels, allowing enough thrombin, a coagulant, to develop to prevent bleeding. It can be used to reduce bleeding episodes in both hemophilia A and hemophilia B since it does not rely on replacing a specific clotting protein, such as factor VIII(8) or factor IX(9). RNA interference is now in phase 3 clinical studies.
Novel non-replacement therapies for hemophilia A and B work by inhibiting the major natural anticoagulant antithrombin, which functions by inactivating activated factor X (FX) and thrombin. The suppression of antithrombin production by hepatocytes is accomplished by the use of RNA interference (RNAi), a natural method of gene silencing found in plants and humans. Fitusiran is an experimental small interfering RNA (siRNA) that inhibits antithrombin production in hepatocytes, restoring adequate hemostasis in people with hemophilia A or B. Fitusiran is now being studied in individuals with or without inhibitors as prophylaxis given subcutaneously every other month or once a month.
Another hemophilia therapy involves interfering with activated protein C (aPC). The thrombin-thrombomodulin complex activates PC to generate aPC, which inactivates FVIIIa and FVa de collaboration with its cofactor protein S, preventing further thrombin generation. These cause the prothrombinase and tenase-complex activities to be inactivated.
Inhibiting aPC may help restore hemostatic equilibrium in hemophilia patients by delaying thrombin production. SerpinPC is an aPC inhibitor that is now being tested in a proof-of-concept experiment in patients with severe hemophilia A and B. This modified serpin forms a covalent connection with aPC but not with a nonactivated PC and does not block thrombin. The absence of reported thrombotic events in clinical trials as of yet may be explained by the intact function of secondary aPC. These are key developments bolstering the hemophilia treatment drugs market progress.
According to the latest hemophilia treatment drugs market trends, the recombinant coagulation factor concentrates product segment is expected to dominate the industry during the forecast period. R&D of new recombinant drugs and gene therapies, such as Fitusiran (phase III trials) and valoctocogene roxaparvovec (phase III candidate), is boosting the segment.
According to the latest hemophilia treatment drugs market forecast, North America is anticipated to hold largest share from 2023 to 2031. Rise in prevalence of hemophilia is fueling the market dynamics of the region. According to the Centers for Disease Control and Prevention (CDC), hemophilia A affects one in every 5,000 male births. Increase in funding for hemophilia treatment is boosting the hemophilia treatment drugs industry statistics.
Many Hemophilia Treatment Centers (HTCs) in the U.S. are housed in large university medical and research facilities. More than 100 HTCs are funded by the federal government. Medical specialists at these HTCs give treatment, education, and support. Patients who receive care and treatment at HTCs generally have a lower risk of hospitalization and bleeding problems than those who receive care elsewhere.
The industry in Asia Pacific is projected to grow at the fastest rate during the forecast period. Rise in adoption of recombinant coagulation factor concentrates in developing countries, such as India and China, is augmenting the hemophilia treatment drugs market expansion in the region. The market in Latin America is poised to grow at a moderate pace during the forecast period.
Most hemophilia treatment drug companies are investing in R&D activities to expand their product portfolio. They are also adopting partnership, collaboration, and M&A strategies to increase their hemophilia treatment drugs market share.
Pfizer Inc., CSL Behring, Kedrion, Takeda Pharmaceutical Company Limited, Novo Nordisk A/S, Bayer AG, F. Hoffmann-La Roche Ltd., Octapharma AG, Biotest AG, and Sanofi S.A. are key entities operating in this market.
These companies have been profiled in the hemophilia treatment drugs market report based on parameters such as company overview, business strategies, financial overview, product portfolio, and business segments.
| Attribute | Detail |
|---|---|
| Market Value in 2022 (Base Year) | US$ 11.8 Bn |
| Market Forecast Value in 2031 | US$ 18.6 Bn |
| Growth Rate (CAGR) | 5.0% |
| Forecast Period | 2023-2031 |
| Historical Data Available for | 2020-2021 |
| Quantitative Units | US$ Bn for Value |
| Market Analysis | Qualitative analysis includes drivers, restraints, opportunities, key trends, key market indicators, Porter’s Five Forces analysis, value chain analysis, and SWOT analysis. Furthermore, at the regional level, the qualitative analysis includes key trends, price trends, and key supplier analysis. |
| Competition Landscape |
|
| Format | Electronic (PDF) + Excel |
| Regions Covered |
|
| Countries Covered |
|
| Market Segmentation |
|
| Companies Profiled |
|
| Customization Scope | Available upon Request |
| Pricing | Available upon Request |
It was valued at US$ 11.8 Bn in 2022
It is likely to grow at a CAGR of 5.0% from 2023 to 2031
R&D of new medications and rise in demand for long-acting novel non-replacement therapies
The recombinant coagulation factor concentrates product segment held largest share in 2022
North America held largest share in 2022
Pfizer Inc., CSL Behring, Kedrion, Takeda Pharmaceutical Company Limited, Novo Nordisk A/S, Bayer AG, F. Hoffmann-La Roche Ltd., Octapharma AG, Biotest AG, and Sanofi S.A.
1. Executive Summary
1.1. Market Definition and Scope
1.2. Market Segmentation
1.3. Key Research Objectives
1.4. Research Highlights
2. Assumptions and Research Methodology
3. Executive Summary: Global Hemophilia Treatment Drugs Market
4. Market Overview
4.1. Market Segmentation
4.1.1. Segment Definition
4.1.2. Industry Evolution / Developments
4.2. Overview
4.3. Market Dynamics
4.3.1. Drivers
4.3.2. Restraints
4.3.3. Opportunities
4.4. Global Hemophilia Treatment Drugs Market Analysis and Forecast, 2023-2031
5. Key Insights
5.1. Pipeline Analysis
5.2. List of Types of Hemophilia Treatment Drugs
5.3. Disease Prevalence & Incidence Rate Globally with Key Countries
5.4. COVID-19 Pandemic Impact on Industry
6. Global Hemophilia Treatment Drugs Market Analysis and Forecast, by Product
6.1. Introduction and Definitions
6.2. Key Findings/Developments
6.3. Market Value Forecast, by Product, 2023–2031
6.3.1. Recombinant Coagulation Factor Concentrates
6.3.1.1. Factor VIII
6.3.1.2. Factor IX
6.3.1.3. Others
6.3.2. Plasma-derived Coagulation Factor Concentrates
6.3.2.1. Factor VIII
6.3.2.2. Factor IX
6.3.2.3. Others
6.3.3. Others
6.4. Market Attractiveness, by Product
7. Global Hemophilia Treatment Drugs Market Analysis and Forecast, by Disease Indication
7.1. Introduction and Definitions
7.2. Key Findings/Developments
7.3. Market Value Forecast, by Disease Indication, 2023–2031
7.3.1. Hemophilia A
7.3.2. Hemophilia B
7.3.3. Others
7.4. Market Attractiveness, by Disease Indication
8. Global Hemophilia Treatment Drugs Market Analysis and Forecast, by Distribution Channel
8.1. Introduction and Definitions
8.2. Key Findings/Developments
8.3. Market Value Forecast, by Distribution Channel, 2023–2031
8.3.1. Hospital Pharmacies
8.3.2. Retail Pharmacies
8.3.3. Online Pharmacies
8.4. Market Attractiveness, by Distribution Channel
9. Global Hemophilia Treatment Drugs Market Analysis and Forecast, by Region
9.1. Key Findings
9.2. Market Value Forecast, by Region, 2023–2031
9.2.1. North America
9.2.2. Europe
9.2.3. Asia Pacific
9.2.4. Latin America
9.2.5. Middle East & Africa
9.3. Market Attractiveness, by Region
10. North America Hemophilia Treatment Drugs Market Analysis and Forecast
10.1. Introduction
10.1.1. Key Findings
10.2. Market Value Forecast, by Product, 2023–2031
10.2.1. Recombinant Coagulation Factor Concentrates
10.2.1.1. Factor VIII
10.2.1.2. Factor IX
10.2.1.3. Others
10.2.2. Plasma-derived Coagulation Factor Concentrates
10.2.2.1. Factor VIII
10.2.2.2. Factor IX
10.2.2.3. Others
10.2.3. Others
10.3. Market Attractiveness, by Product
10.4. Market Value Forecast, by Disease Indication, 2023–2031
10.4.1. Hemophilia A
10.4.2. Hemophilia B
10.4.3. Others
10.5. Market Attractiveness, by Disease Indication
10.6. Market Value Forecast, by Distribution Channel, 2023–2031
10.6.1. Hospital Pharmacies
10.6.2. Retail Pharmacies
10.6.3. Online Pharmacies
10.7. Market Attractiveness, by Distribution Channel
10.8. Market Value Forecast, by Country, 2023–2031
10.8.1. U.S.
10.8.2. Canada
10.9. Market Attractiveness Analysis
10.9.1. By Product
10.9.2. By Disease Indication
10.9.3. By Distribution Channel
10.9.4. By Country
11. Europe Hemophilia Treatment Drugs Market Analysis and Forecast
11.1. Introduction
11.1.1. Key Findings
11.1.2. Recombinant Coagulation Factor Concentrates
11.1.2.1. Factor VIII
11.1.2.2. Factor IX
11.1.2.3. Others
11.1.3. Plasma-derived Coagulation Factor Concentrates
11.1.3.1. Factor VIII
11.1.3.2. Factor IX
11.1.3.3. Others
11.1.4. Others
11.2. Market Attractiveness, by Product
11.3. Market Value Forecast, by Disease Indication, 2023–2031
11.3.1. Hemophilia A
11.3.2. Hemophilia B
11.3.3. Others
11.4. Market Attractiveness, by Disease Indication
11.5. Market Value Forecast, by Distribution Channel, 2023–2031
11.5.1. Hospital Pharmacies
11.5.2. Retail Pharmacies
11.5.3. Online Pharmacies
11.6. Market Attractiveness, by Distribution Channel
11.7. Market Value Forecast, by Country/Sub-region, 2023–2031
11.7.1. Germany
11.7.2. U.K.
11.7.3. France
11.7.4. Italy
11.7.5. Spain
11.7.6. Rest of Europe
11.8. Market Attractiveness Analysis
11.8.1. By Product
11.8.2. By Disease Indication
11.8.3. By Distribution Channel
11.8.4. By Country/Sub-region
12. Asia Pacific Hemophilia Treatment Drugs Market Analysis and Forecast
12.1. Introduction
12.1.1. Key Findings
12.2. Market Value Forecast, by Product, 2023–2031
12.2.1. Recombinant Coagulation Factor Concentrates
12.2.1.1. Factor VIII
12.2.1.2. Factor IX
12.2.1.3. Others
12.2.2. Plasma-derived Coagulation Factor Concentrates
12.2.2.1. Factor VIII
12.2.2.2. Factor IX
12.2.2.3. Others
12.2.3. Others
12.3. Market Attractiveness, by Product
12.4. Market Value Forecast, by Disease Indication, 2023–2031
12.4.1. Hemophilia A
12.4.2. Hemophilia B
12.4.3. Others
12.5. Market Attractiveness, by Disease Indication
12.6. Market Value Forecast, by Distribution Channel, 2023–2031
12.6.1. Hospital Pharmacies
12.6.2. Retail Pharmacies
12.6.3. Online Pharmacies
12.7. Market Attractiveness, by Distribution Channel
12.8. Market Value Forecast, by Country/Sub-region, 2023–2031
12.8.1. China
12.8.2. Japan
12.8.3. India
12.8.4. Australia & New Zealand
12.8.5. Rest of Asia Pacific
12.9. Market Attractiveness Analysis
12.9.1. By Product
12.9.2. By Disease Indication
12.9.3. By Distribution Channel
12.9.4. By Country/Sub-region
13. Latin America Hemophilia Treatment Drugs Market Analysis and Forecast
13.1. Introduction
13.1.1. Key Findings
13.2. Market Value Forecast, by Product, 2023–2031
13.2.1. Recombinant Coagulation Factor Concentrates
13.2.1.1. Factor VIII
13.2.1.2. Factor IX
13.2.1.3. Others
13.2.2. Plasma-derived Coagulation Factor Concentrates
13.2.2.1. Factor VIII
13.2.2.2. Factor IX
13.2.2.3. Others
13.2.3. Others
13.3. Market Attractiveness, by Product
13.4. Market Value Forecast, by Disease Indication, 2023–2031
13.4.1. Hemophilia A
13.4.2. Hemophilia B
13.4.3. Others
13.5. Market Attractiveness, by Disease Indication
13.6. Market Value Forecast, by Distribution Channel, 2023–2031
13.6.1. Hospital Pharmacies
13.6.2. Retail Pharmacies
13.6.3. Online Pharmacies
13.7. Market Attractiveness, by Distribution Channel
13.8. Market Value Forecast, by Country/Sub-region, 2023–2031
13.8.1. Brazil
13.8.2. Mexico
13.8.3. Rest of Latin America
13.9. Market Attractiveness Analysis
13.9.1. By Product
13.9.2. By Disease Indication
13.9.3. By Distribution Channel
13.9.4. By Country/Sub-region
14. Middle East & Africa Hemophilia Treatment Drugs Market Analysis and Forecast
14.1. Introduction
14.1.1. Key Findings
14.2. Market Value Forecast, by Product, 2023–2031
14.2.1. Recombinant Coagulation Factor Concentrates
14.2.1.1. Factor VIII
14.2.1.2. Factor IX
14.2.1.3. Others
14.2.2. Plasma-derived Coagulation Factor Concentrates
14.2.2.1. Factor VIII
14.2.2.2. Factor IX
14.2.2.3. Others
14.2.3. Others
14.3. Market Attractiveness, by Product
14.4. Market Value Forecast, by Disease Indication, 2023–2031
14.4.1. Hemophilia A
14.4.2. Hemophilia B
14.4.3. Others
14.5. Market Attractiveness, by Disease Indication
14.6. Market Value Forecast, by Distribution Channel, 2023–2031
14.6.1. Hospital Pharmacies
14.6.2. Retail Pharmacies
14.6.3. Online Pharmacies
14.7. Market Attractiveness, by Distribution Channel
14.8. Market Value Forecast, by Country/Sub-region, 2023–2031
14.8.1. GCC Countries
14.8.2. South Africa
14.8.3. Rest of Middle East & Africa
14.9. Market Attractiveness Analysis
14.9.1. By Product
14.9.2. By Disease Indication
14.9.3. By Distribution Channel
14.9.4. By Country/Sub-region
15. Competition Landscape
15.1. Market Player – Competition Matrix (By Tier and Size of Companies)
15.2. Market Share Analysis, by Company (2022)
15.3. Company Profiles
15.3.1. Pfizer Inc.
15.3.1.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.1.2. Product Portfolio
15.3.1.3. Financial Overview
15.3.1.4. SWOT Analysis
15.3.1.5. Strategic Overview
15.3.2. CSL Behring
15.3.2.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.2.2. Product Portfolio
15.3.2.3. Financial Overview
15.3.2.4. SWOT Analysis
15.3.2.5. Strategic Overview
15.3.3. Kedrion
15.3.3.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.3.2. Product Portfolio
15.3.3.3. Financial Overview
15.3.3.4. SWOT Analysis
15.3.3.5. Strategic Overview
15.3.4. Takeda Pharmaceutical Company Limited
15.3.4.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.4.2. Product Portfolio
15.3.4.3. Financial Overview
15.3.4.4. SWOT Analysis
15.3.4.5. Strategic Overview
15.3.5. Novo Nordisk A/S
15.3.5.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.5.2. Product Portfolio
15.3.5.3. Financial Overview
15.3.5.4. SWOT Analysis
15.3.5.5. Strategic Overview
15.3.6. Bayer AG
15.3.6.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.6.2. Product Portfolio
15.3.6.3. Financial Overview
15.3.6.4. SWOT Analysis
15.3.6.5. Strategic Overview
15.3.7. F. Hoffmann-La Roche Ltd.
15.3.7.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.7.2. Product Portfolio
15.3.7.3. Financial Overview
15.3.7.4. SWOT Analysis
15.3.7.5. Strategic Overview
15.3.8. Octapharma AG
15.3.8.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.8.2. Product Portfolio
15.3.8.3. Financial Overview
15.3.8.4. SWOT Analysis
15.3.8.5. Strategic Overview
15.3.9. Biotest AG
15.3.9.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.9.2. Product Portfolio
15.3.9.3. Financial Overview
15.3.9.4. SWOT Analysis
15.3.9.5. Strategic Overview
15.3.10. Sanofi S.A.
15.3.10.1. Company Overview (HQ, Business Segments, Employee Strength)
15.3.10.2. Product Portfolio
15.3.10.3. Financial Overview
15.3.10.4. SWOT Analysis
15.3.10.5. Strategic Overview
List of Tables
Table 1: Global Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Product, 2023–2031
Table 2: Global Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Disease Indication, 2023–2031
Table 3: Global Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Distribution Channel, 2023–2031
Table 4: Global Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Region, 2023–2031
Table 5: North America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Country, 2023–2031
Table 6: North America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Product, 2023–2031
Table 7: North America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Disease Indication, 2023–2031
Table 8: North America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Distribution Channel, 2023–2031
Table 9: Europe Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Country/Sub-region, 2023–2031
Table 10: Europe Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Product, 2023–2031
Table 11: Europe Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Disease Indication, 2023–2031
Table 12: Europe Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Distribution Channel 2023–2031
Table 13: Asia Pacific Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Country/Sub-region, 2023–2031
Table 14: Asia Pacific Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Product, 2023–2031
Table 15: Asia Pacific Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Disease Indication, 2023–2031
Table 16: Asia Pacific Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Distribution Channel, 2023–2031
Table 17: Latin America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Country/Sub-region, 2023–2031
Table 18: Latin America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Product, 2023–2031
Table 19: Latin America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Disease Indication, 2023–2031
Table 20: Latin America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Distribution Channel 2023–2031
Table 21: Middle East & Africa Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Country/Sub-region, 2023–2031
Table 22: Middle East & Africa Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Product, 2023–2031
Table 23: Middle East & Africa Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Disease Indication, 2023–2031
Table 24: Middle East & Africa Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, by Distribution Channel 2023–2031
List of Figures
Figure 1: Global Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, 2023–2031
Figure 2: Global Hemophilia Treatment Drugs Market Value Share, by Product, 2022
Figure 3: Global Hemophilia Treatment Drugs Market Value Share, by Disease Indication, 2022
Figure 4: Global Hemophilia Treatment Drugs Market Value Share, by Distribution Channel, 2022
Figure 5: Global Hemophilia Treatment Drugs Market Value Share Analysis, by Product, 2022 and 2031
Figure 6: Global Hemophilia Treatment Drugs Market Attractiveness Analysis, by Product, 2023–2031
Figure 7: Global Hemophilia Treatment Drugs Market Value Share Analysis, by Disease Indication, 2022 and 2031
Figure 8: Global Hemophilia Treatment Drugs Market Attractiveness Analysis, by Disease Indication, 2023–2031
Figure 9: Global Hemophilia Treatment Drugs Market Value Share Analysis, by Distribution Channel, 2022 and 2031
Figure 10: Global Hemophilia Treatment Drugs Market Attractiveness Analysis, by Distribution Channel 2023–2031
Figure 11: Global Hemophilia Treatment Drugs Market Value Share Analysis, by Region, 2022 and 2031
Figure 12: Global Hemophilia Treatment Drugs Market Attractiveness Analysis, by Region, 2023–2031
Figure 13: North America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, 2023–2031
Figure 14: North America Hemophilia Treatment Drugs Market Value Share Analysis, by Country, 2022 and 2031
Figure 15: North America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Country, 2023–2031
Figure 16: North America Hemophilia Treatment Drugs Market Value Share Analysis, by Product, 2022 and 2031
Figure 17: North America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Product, 2023–2031
Figure 18: North America Hemophilia Treatment Drugs Market Value Share Analysis, by Disease Indication, 2022 and 2031
Figure 19: North America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Disease Indication, 2023–2031
Figure 20: North America Hemophilia Treatment Drugs Market Value Share Analysis, by Distribution Channel, 2022 and 2031
Figure 21: North America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Distribution Channel 2023–2031
Figure 22: Europe Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, 2023–2031
Figure 23: Europe Hemophilia Treatment Drugs Market Value Share Analysis, by Country/Sub-region, 2022 and 2031
Figure 24: Europe Hemophilia Treatment Drugs Market Attractiveness Analysis, by Country/Sub-region, 2023–2031
Figure 25: Europe Hemophilia Treatment Drugs Market Value Share Analysis, by Product, 2022 and 2031
Figure 26: Europe America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Product, 2023–2031
Figure 27: Europe Hemophilia Treatment Drugs Market Value Share Analysis, by Disease Indication, 2022 and 2031
Figure 28: Europe Hemophilia Treatment Drugs Market Attractiveness Analysis, by Disease Indication, 2023–2031
Figure 29: Europe Hemophilia Treatment Drugs Market Value Share Analysis, by Distribution Channel, 2022 and 2031
Figure 30: Europe Hemophilia Treatment Drugs Market Attractiveness Analysis, by Distribution Channel 2023–2031
Figure 31: Asia Pacific Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, 2023–2031
Figure 32: Asia Pacific Hemophilia Treatment Drugs Market Value Share Analysis, by Country/Sub-region, 2022 and 2031
Figure 33: Asia Pacific Hemophilia Treatment Drugs Market Attractiveness Analysis, by Country/Sub-region, 2023–2031
Figure 34: Asia Pacific Hemophilia Treatment Drugs Market Value Share Analysis, by Product, 2022 and 2031
Figure 35: Asia Pacific Hemophilia Treatment Drugs Market Attractiveness Analysis, by Product, 2023–2031
Figure 36: Asia Pacific Hemophilia Treatment Drugs Market Value Share Analysis, by Disease Indication, 2022 and 2031
Figure 37: Asia Pacific Hemophilia Treatment Drugs Market Attractiveness Analysis, by Disease Indication, 2023–2031
Figure 38: Asia Pacific Hemophilia Treatment Drugs Market Value Share Analysis, by Distribution Channel, 2022 and 2031
Figure 39: Asia Pacific Hemophilia Treatment Drugs Market Attractiveness Analysis, by Distribution Channel 2023–2031
Figure 40: Latin America Hemophilia Treatment Drugs Market Value (US$ Bn) Forecast, 2023–2031
Figure 41: Latin America Hemophilia Treatment Drugs Market Value Share Analysis, by Country/Sub-region, 2022 and 2031
Figure 42: Latin America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Country/Sub-region, 2023–2031
Figure 43: Latin America Hemophilia Treatment Drugs Market Value Share Analysis, by Product, 2022 and 2031
Figure 44: Latin America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Product, 2023–2031
Figure 45: Latin America Hemophilia Treatment Drugs Market Value Share Analysis, by Disease Indication, 2022 and 2031
Figure 46: Latin America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Disease Indication, 2023–2031
Figure 47: Latin America Hemophilia Treatment Drugs Market Value Share Analysis, by Distribution Channel, 2022 and 2031
Figure 48: Latin America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Distribution Channel, 2023–2031
Figure 49: Middle East & Africa Hemophilia Treatment Drugs Market Value Share Analysis, by Country/Sub-region, 2022 and 2031
Figure 50: Middle East & Africa Hemophilia Treatment Drugs Market Attractiveness Analysis, by Country/Sub-region, 2023–2031
Figure 51: Middle East & Africa America Hemophilia Treatment Drugs Market Value Share Analysis, by Product, 2023–2031
Figure 52: Middle East & Africa America Hemophilia Treatment Drugs Market Attractiveness Analysis, by Product, 2023–2031
Figure 53: Middle East & Africa Hemophilia Treatment Drugs Market Value Share Analysis, by Distribution Channel, 2022 and 2031
Figure 54: Middle East & Africa Hemophilia Treatment Drugs Market Attractiveness Analysis, by Distribution Channel 2023–2031
Figure 55: Global Hemophilia Treatment Drugs Market Share Analysis, by Company (2022)