Reports
Reports
Technological advancements have played an imperative role in transforming the modern day healthcare industry. Over the past decade, minimally invasive surgeries, also known as laparoscopy, have garnered significance, as they require smaller incisions. With additional efforts toward minimizing the size of surgical incisions, minimally invasive procedures are set to gain traction in the near future. The advent of robotic technology, significant developments in endomechanical devices, and adoption of cutting-edge optical camera and imaging technologies are projected to accelerate the growth of the endosurgery devices market during the forecast period (2019-2027).
The demand for laparoscopic procedures is witnessing a notable growth across the world, owing to a range of benefits, such as less morbidity, faster healing, swift recovery, improved outcome, and low degree of discomfort. Surge in the adoption of these procedures has directly impacted the demand for endosurgery devices. Moreover, growing awareness regarding the advantages of laparoscopic surgeries, advent of robotic assisted endosurgery devices, and evolution of modern day surgical procedures are some of the leading factors that are expected to propel the expansion of the endosurgery devices market in the near future.
Minimally invasive procedures are associated with shorter hospital stays and significantly low degree of discomfort compared to conventional surgical techniques. However, despite these advantages, alternative treatments such as technologically advanced endoscopic surgeries, which include natural orifice transluminal endoscopic surgery (NOTES), endoscopic sub mucosal dissection (ESD), and suturing that require training and a high level of expertise could slow down the growth of the endosurgery devices market.
Although a broad range of endosurgery devices, such electrosurgical knifes, hemostatic forceps, suturing devices, and distal attachment caps are available, these conventional endosurgery devices lack the capabilities of advanced endosurgery devices. For instance, there is a dearth of desired dexterity required to carry out fine surgical maneuvers such nonaxial tissue manipulation and triangulation of instruments. Due to the limitations of conventional endosurgery devices, endoscopic robotic systems have made an entry into the endosurgery devices market. The entry of robotic endosurgery devices, including dissection tools, electrocautery devices, and sutures have resulted in effective, precise, reliable, and safe endoscopic procedures. At present, manufacturers operating in the endosurgery devices market are focusing on addressing the issues pertaining to instrument control and locomotion to improve the performance and overall output of these devices.
Participants within the endosurgery devices sphere are investing resources on the development of robotic systems with improved flexibility. For instance, the Flex Robotic System manufactured by Medrobotics was specially developed to perform neck and head surgeries. It was given the U.S. FDA approval in 2017. The adoption of these devices is expected to gain steady momentum in the coming years, as these devices continue to simplify the complexities involved with conventional procedures.
Sensing the growing demand for advanced endosurgery devices, stakeholders in the current endosurgery devices market landscape are leaning toward the development of new, cutting-edge endosurgery devices and are simultaneously pushing to gain approval from the FDA. For instance, in September 2019, Apollo Endosurgery was given the FDA clearance for the newly developed suture anchor device specifically designed to work in conjunction with OverStitch endoscopic suturing systems. The company revealed that obtaining the clearance from the FDA would enable it to improve the overall distribution of their suture-anchor systems and gross margins. Moreover, with better control over their supply chain, the company aims to expand its footprint in the global endosurgery devices market.
In September 2019, Ethicon launched the first powered circular stapler to address the complications related to gastric, thoracic, and colorectal surgery. The newly developed endosurgery device, ECHELON CIRCULAR Powered Stapler is a combination of two novel technologies of the company –3D Stapling Technology and Gripping Surface Technology (GST). Companies operating in the healthcare technology sphere are expected to focus on the development of cutting-edge devices that offer high degree of precision, accuracy, and end results.
Analysts’ Viewpoint
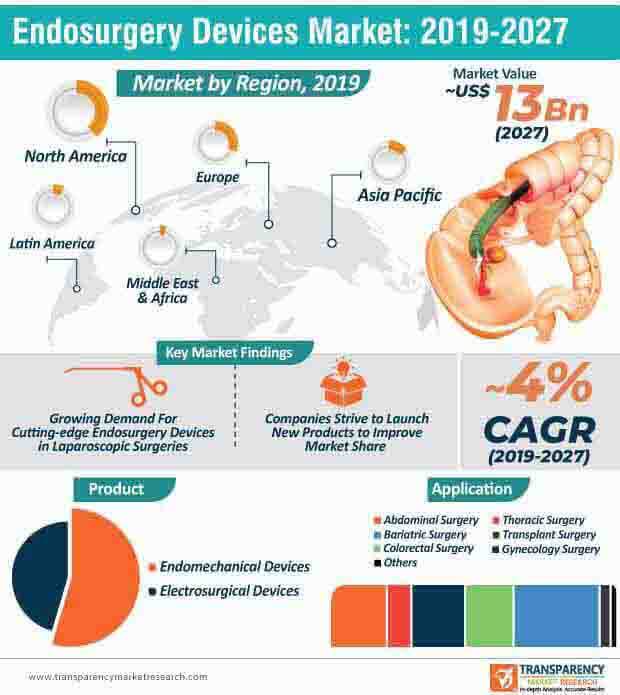
The endosurgery devices market is expected to grow at a CAGR of ~4% during the forecast period and attain a market value of ~US$ 13 Bn by 2027. The onset of state-of-the-art technologies, developments in robotic surgical systems, and integration of advanced imaging technologies will continue to bolster the growth of the market for endosurgery devices in the near future. Companies should engage in the development of new products with improved capabilities and functionalities. The adoption of these devices for abdominal, thoracic, bariatric, and gynecology surgeries, among others, will continue to grow during the forecast period, particularly in developed regions, including North America and Europe.
Endosurgery Devices Market: Overview
Launch of New Endosurgery Devices-based Products: Key Drivers
Rise in Minimally Invasive Surgeries
Surge in Adoption of Advanced Robotic Endosurgery Devices
High Demand for Endosurgery Devices in Bariatric Surgery
North America to Dominate Endosurgery Devices Market
Endosurgery Devices Market: Competition Landscape
In the global endosurgery devices market report, we have discussed individual strategies, followed by company profiles of the manufacturers of endosurgery devices. The ‘competitive landscape’ section has been included in the global endosurgery devices market report to provide readers with a dashboard view of the key players operating in the global endosurgery devices market.
Endosurgery devices market to reach a valuation of ~US$13 Bn By 2027
Endosurgery devices market is projected to expand at a CAGR of ~4% from 2019 to 2027
Endosurgery devices market is driven by growing awareness regarding the advantages of laparoscopic surgeries
North America accounted for a major share of the global endosurgery devices market
Key players in the global endosurgery devices market are Medtronic, Stryker, Ethicon, Inc., KARL STORZ SE & Co. KG, Olympus Corporation
1. Preface
1.1. Market Definition and Scope
1.2. Market Segmentation
1.3. Key Research Objectives
1.4. Research Highlights
2. Assumptions and Research Methodology
3. Executive Summary: Global Endosurgery Devices Market
4. Market Overview
4.1. Introduction
4.1.1. Product Definition
4.2. Overview
4.3. Market Dynamics
4.3.1. Drivers
4.3.2. Restraints
4.3.3. Opportunities
4.4. Global Endosurgery Devices Market Analysis and Forecast, 2019–2027
5. Market Outlook
5.1. Technological Advancements
5.2. Key Mergers & Acquisitions
5.3. Applied SWOT Analysis
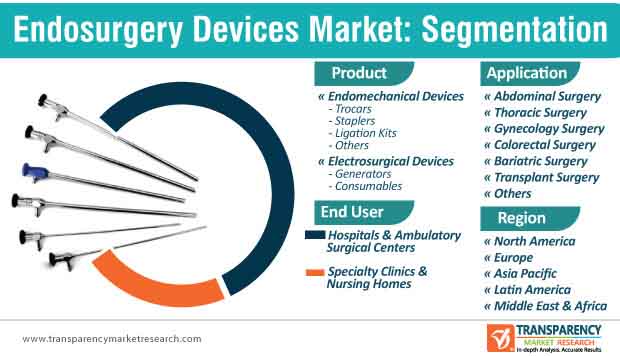
6. Global Endosurgery Devices Market Analysis and Forecast, by Product
6.1. Introduction & Definition
6.2. Market Value Forecast, by Product, 2019–2027
6.2.1. Endomechanical Devices
6.2.1.1. Trocars
6.2.1.2. Staplers
6.2.1.3. Ligation Kits
6.2.1.4. Others
6.2.2. Electrosurgical Devices
6.2.2.1. Generators
6.2.2.2. Consumables
6.2.2.2.1. Monopolar
6.2.2.2.2. Bipolar
6.2.2.2.3. Vessel Sealing
6.3. Market Attractiveness, by Product
7. Global Endosurgery Devices Market Analysis and Forecast, by Application
7.1. Introduction & Definition
7.2. Market Value Forecast, by Application, 2019–2027
7.2.1. Abdominal Surgery
7.2.2. Thoracic Surgery
7.2.3. Gynecology
7.2.4. Colorectal Surgery
7.2.5. Bariatric Surgery
7.2.6. Transplant Surgery
7.2.7. Others
7.3. Market Attractiveness, by Application
8. Global Endosurgery Devices Market Analysis and Forecast, by End-user
8.1. Introduction & Definition
8.2. Market Value Forecast, by End-user, 2019–2027
8.2.1. Hospitals & Ambulatory Surgical Centers
8.2.2. Specialty Clinics & Nursing Homes
8.3. Market Attractiveness, by End-user
9. Global Endosurgery Devices Market Analysis and Forecast, by Region
9.1. Global Endosurgery Devices Market Scenario
9.2. Market Value Forecast, by Region, 2019–2027
9.2.1. North America
9.2.2. Europe
9.2.3. Asia Pacific
9.2.4. Latin America
9.2.5. Middle East & Africa
9.3. Market Attractiveness By Region
10. North America Endosurgery Devices Market Analysis and Forecast
10.1. Market Overview
10.2. Market Value Forecast, by Product, 2019–2027
10.2.1. Endomechanical Devices
10.2.1.1. Trocars
10.2.1.2. Staplers
10.2.1.3. Ligation Kits
10.2.1.4. Others
10.2.2. Electrosurgical Devices
10.2.2.1. Generators
10.2.2.2. Consumables
10.2.2.2.1. Monopolar
10.2.2.2.2. Bipolar
10.2.2.2.3. Vessel Sealing
10.3. Market Value Forecast, by Application, 2019–2027
10.3.1. Abdominal Surgery
10.3.2. Thoracic Surgery
10.3.3. Gynecology
10.3.4. Colorectal Surgery
10.3.5. Bariatric Surgery
10.3.6. Transplant Surgery
10.3.7. Others
10.4. Market Value Forecast, by End-user, 2019–2027
10.4.1. Hospitals & Ambulatory Surgical Centers
10.4.2. Specialty Clinics & Nursing Homes
10.5. Market Value Forecast, by Country, 2019–2027
10.5.1. U.S.
10.5.2. Canada
10.6. Market Attractiveness Analysis
10.6.1. By Product
10.6.2. By Application
10.6.3. By End-user
10.6.4. By Country
11. Europe Endosurgery Devices Market Analysis and Forecast
11.1. Market Overview
11.2. Market Value Forecast, by Product, 2019–2027
11.2.1. Endomechanical Devices
11.2.1.1. Trocars
11.2.1.2. Staplers
11.2.1.3. Ligation Kits
11.2.1.4. Others
11.2.2. Electrosurgical Devices
11.2.2.1. Generators
11.2.2.2. Consumables
11.2.2.2.1. Monopolar
11.2.2.2.2. Bipolar
11.2.2.2.3. Vessel Sealing
11.3. Market Value Forecast, by Application, 2019–2027
11.3.1. Abdominal Surgery
11.3.2. Thoracic Surgery
11.3.3. Gynecology
11.3.4. Colorectal Surgery
11.3.5. Bariatric Surgery
11.3.6. Transplant Surgery
11.3.7. Others
11.4. Market Value Forecast, by End-user , 2019–2027
11.4.1. Hospitals & Ambulatory Surgical Centers
11.4.2. Specialty Clinics & Nursing Homes
11.5. Market Value Forecast, by Country/Sub-region, 2019–2027
11.5.1. Germany
11.5.2. U.K.
11.5.3. France
11.5.4. Italy
11.5.5. Spain
11.5.6. Rest of Europe
11.6. Market Attractiveness Analysis
11.6.1. By Product
11.6.2. By Application
11.6.3. By End-user
11.6.4. By Country/Sub-Region
12. Asia Pacific Endosurgery Devices Market Analysis and Forecast
12.1. Market Overview
12.2. Market Value Forecast, by Product, 2019–2027
12.2.1. Endomechanical Devices
12.2.1.1. Trocars
12.2.1.2. Staplers
12.2.1.3. Ligation Kits
12.2.1.4. Others
12.2.2. Electrosurgical Devices
12.2.2.1. Generators
12.2.2.2. Consumables
12.2.2.2.1. Monopolar
12.2.2.2.2. Bipolar
12.2.2.2.3. Vessel Sealing
12.3. Market Value Forecast, by Application, 2019–2027
12.3.1. Abdominal Surgery
12.3.2. Thoracic Surgery
12.3.3. Gynecology
12.3.4. Colorectal Surgery
12.3.5. Bariatric Surgery
12.3.6. Transplant Surgery
12.3.7. Others
12.4. Market Value Forecast, by End-user, 2019–2027
12.4.1. Hospitals & Ambulatory Surgical Centers
12.4.2. Specialty Clinics & Nursing Homes
12.5. Market Value Forecast, by Country/Sub-region, 2019–2027
12.5.1. Japan
12.5.2. China
12.5.3. India
12.5.4. Australia & New Zealand
12.5.5. Rest of Asia Pacific
12.6. Market Attractiveness Analysis
12.6.1. By Product
12.6.2. By Application
12.6.3. By End-user
12.6.4. By Country/Sub-Region
13. Latin America Endosurgery Devices Market Analysis and Forecast
13.1. Market Overview
13.2. Market Value Forecast, by Product, 2019–2027
13.2.1. Endomechanical Devices
13.2.1.1. Trocars
13.2.1.2. Staplers
13.2.1.3. Ligation Kits
13.2.1.4. Others
13.2.2. Electrosurgical Devices
13.2.2.1. Generators
13.2.2.2. Consumables
13.2.2.2.1. Monopolar
13.2.2.2.2. Bipolar
13.2.2.2.3. Vessel Sealing
13.3. Market Value Forecast, by Application, 2019–2027
13.3.1. Abdominal Surgery
13.3.2. Thoracic Surgery
13.3.3. Gynecology
13.3.4. Colorectal Surgery
13.3.5. Bariatric Surgery
13.3.6. Transplant Surgery
13.3.7. Others
13.4. Market Value Forecast, by End-user, 2019–2027
13.4.1. Hospitals & Ambulatory Surgical Centers
13.4.2. Specialty Clinics & Nursing Homes
13.5. Market Value Forecast, by Country/Sub-region, 2019–2027
13.5.1. Brazil
13.5.2. Mexico
13.5.3. Rest of Latin America
13.6. Market Attractiveness Analysis
13.6.1. By Product
13.6.2. By Application
13.6.3. By End-user
13.6.4. By Country/Sub-Region
14. Middle East & Africa Endosurgery Devices Market Analysis and Forecast
14.1. Market Overview
14.2. Market Value Forecast, by Product, 2019–2027
14.2.1. Endomechanical Devices
14.2.1.1. Trocars
14.2.1.2. Staplers
14.2.1.3. Ligation Kits
14.2.1.4. Others
14.2.2. Electrosurgical Devices
14.2.2.1. Generators
14.2.2.2. Consumables
14.2.2.2.1. Monopolar
14.2.2.2.2. Bipolar
14.2.2.2.3. Vessel Sealing
14.3. Market Value Forecast, by Application, 2019–2027
14.3.1. Abdominal Surgery
14.3.2. Thoracic Surgery
14.3.3. Gynecology
14.3.4. Colorectal Surgery
14.3.5. Bariatric Surgery
14.3.6. Transplant Surgery
14.3.7. Others
14.4. Market Value Forecast, by End-user, 2019–2027
14.4.1. Hospitals & Ambulatory Surgical Centers
14.4.2. Specialty Clinics & Nursing Homes
14.5. Market Value Forecast By Country/Sub-region, 2019–2027
14.5.1. GCC Countries
14.5.2. South Africa
14.5.3. Israel
14.5.4. Rest of Middle East & Africa
14.6. Market Attractiveness Analysis
14.6.1. By Product
14.6.2. By Application
14.6.3. By End-user
14.6.4. By Country/Sub-Region
15. Competition Landscape
15.1. Market Player - Competition Matrix
15.2. Market Position Analysis/Ranking, by Company, 2018
15.3. Company Profiles
15.3.1. Medtronic
15.3.1.1. Company Description
15.3.1.2. Product Portfolio
15.3.1.3. Financial Overview
15.3.1.4. Strategic Overview
15.3.1.5. SWOT Analysis
15.3.2. Stryker
15.3.2.1. Company Description
15.3.2.2. Financial Overview
15.3.2.3. Strategic Overview
15.3.2.4. SWOT Analysis
15.3.3. Ethicon, Inc. (Johnson & Johnson)
15.3.3.1. Company Description
15.3.3.2. Product Portfolio
15.3.3.3. Financial Overview
15.3.3.4. Strategic Overview
15.3.3.5. SWOT Analysis
15.3.4. Karl Storz SE & Co. KG
15.3.4.1. Company Description
15.3.4.2. Strategic Overview
15.3.4.3. SWOT Analysis
15.3.5. Olympus Corporation
15.3.5.1. Company Description
15.3.5.2. Financial Overview
15.3.5.3. Strategic Overview
15.3.5.4. SWOT Analysis
15.3.6. Erbe Elektromedizin GmbH
15.3.6.1. Company Description
15.3.6.2. Strategic Overview
15.3.6.3. SWOT Analysis
15.3.7. Meril Life Sciences Pvt. Ltd.
15.3.7.1. Company Description
15.3.7.2. Product Portfolio
15.3.7.3. Strategic Overview
15.3.7.4. SWOT Analysis
15.3.8. Healthium Medtech
15.3.8.1. Company Description
15.3.8.2. Strategic Overview
15.3.8.3. SWOT Analysis
15.3.9. CONMED Corporation
15.3.9.1. Company Description
15.3.9.2. Product Portfolio
15.3.9.3. Financial Overview
15.3.9.4. Strategic Overview
15.3.9.5. SWOT Analysis
15.3.10. Reach Surgical
15.3.10.1. Company Description
15.3.10.2. Strategic Overview
15.3.10.3. SWOT Analysis
List of Tables
Table 01: Key Mergers & Acquisitions in the Endosurgery Devices Market
Table 02: Global Endosurgery Devices Market Value (US$ Mn) Forecast, by Product, 2017–2027
Table 03: Global Endosurgery Devices Market Value (US$ Mn) Forecast, by Endomechanical Devices, 2017–2027
Table 04: Global Endosurgery Devices Market Value (US$ Mn) Forecast, by Electrosurgical Devices, 2017–2027
Table 05: Global Endosurgery Devices Market Value (US$ Mn) Forecast, by Consumables, 2017–2027
Table 06: Global Endosurgery Devices Market Value (US$ Mn) Forecast, by Application, 2017–2027
Table 07: Global Endosurgery Devices Market Value (US$ Mn) Forecast, by End-user, 2017–2027
Table 08: Global Endosurgery Devices Market Value (US$ Mn) Forecast, by Region, 2017–2027
Table 09: North America Endosurgery Devices Market Value (US$ Mn) Forecast, by Country, 2017–2027
Table 10: North America Endosurgery Devices Market Value (US$ Mn) Forecast, by Product, 2017–2027
Table 11: North America Endosurgery Devices Market Value (US$ Mn) Forecast, by Endomechanical Devices, 2017–2027
Table 12: North America Endosurgery Devices Market Value (US$ Mn) Forecast, by Electrosurgical Devices, 2017–2027
Table 13: North America Endosurgery Devices Market Value (US$ Mn) Forecast, by Consumables, 2017–2027
Table 14: North America Endosurgery Devices Market Value (US$ Mn) Forecast, by Application, 2017–2027
Table 15: North America Endosurgery Devices Market Value (US$ Mn) Forecast, by Product, 2017–2027
Table 16: Europe Endosurgery Devices Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
Table 17: Europe Endosurgery Devices Market Value (US$ Mn) Forecast, by Product, 2017–2027
Table 18: Europe Endosurgery Devices Market Value (US$ Mn) Forecast, by Endomechanical Devices, 2017–2027
Table 19: Europe Endosurgery Devices Market Value (US$ Mn) Forecast, by Electrosurgical Devices, 2017–2027
Table 20: Europe Endosurgery Devices Market Value (US$ Mn) Forecast, by Consumables, 2017–2027
Table 21: Europe Endosurgery Devices Market Value (US$ Mn) Forecast, by Application, 2017–2027
Table 22: Europe Endosurgery Devices Market Value (US$ Mn) Forecast, by End-user, 2017–2027
Table 23: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast, by Country/Sub-Region, 2017–2027
Table 24: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast, by Product, 2017–2027
Table 25: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast, by Endomechanical Devices, 2017–2027
Table 26: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast, by Electrosurgical Devices, 2017–2027
Table 27: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast, by Consumables, 2017–2027
Table 28: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast, by Application, 2017–2027
Table 29: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast, by End-user, 2017–2027
Table 30: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2027
Table 31: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast, by Product, 2017–2027
Table 32: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast, by Endomechanical Devices, 2017–2027
Table 33: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast, by Electrosurgical Devices, 2017–2027
Table 34: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast, by Consumables, 2017–2027
Table 35: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast, by Application, 2017–2027
Table 36: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast, by End-user, 2017–2027
Table 37: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast, by Country/Sub-Region, 2017–2027
Table 38: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast, by Product, 2017–2027
Table 39: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast, by Endomechanical Devices, 2017–2027
Table 40: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast, by Electrosurgical Devices, 2017–2027
Table 41: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast, by Consumables, 2017–2027
Table 42: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast, by Application, 2017–2027
Table 43: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast, by End-user, 2017–2027
List of Figures
Figure 01: Global Endosurgery Devices Market Value (US$ Mn) and Distribution (%), by Region, 2018 and 2027
Figure 02: Global Endosurgery Devices Market Value Share (%), by Application, 2018
Figure 03: Global Endosurgery Devices Market Value Share Analysis by Product, 2018
Figure 04: Global Endosurgery Devices Market Value Share, by Product, 2018
Figure 05: Global Endosurgery Devices Market Value Share, by Application, 2018
Figure 06: Global Endosurgery Devices Market Value Share, by End-user, 2018
Figure 07: Global Endosurgery Devices Value Share, by Region, 2018
Figure 08: Global Endosurgery Devices Market Value (US$ Mn) Forecast, 2017–2027
Figure 09: Global Endosurgery Devices Market Value Share (%), by Product, 2018 and 2027
Figure 10: Global Endosurgery Devices Market Value (US$ Mn) Forecast and Y-o-Y Growth (%) Projection, by Endomechanical Devices, 2017–2027
Figure 11: Global Endosurgery Devices Market Value (US$ Mn) Forecast and Y-o-Y Growth (%) Projection, by Electrosurgical Devices, 2017–2027
Figure 12: Global Endosurgery Devices Market Attractiveness Analysis, by Product, 2019–2027
Figure 13: Global Endosurgery Devices Market Value Share, by Application, 2018 and 2027
Figure 14: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Abdominal Surgery, 2017–2027
Figure 15: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Thoracic Surgery, 2017–2027
Figure 16: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Gynecology Surgery, 2017–2027
Figure 17: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Colorectal Surgery, 2017–2027
Figure 18: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Bariatric Surgery, 2017–2027
Figure 19: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Transplant Surgery, 2017–2027
Figure 20: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Others, 2017–2027
Figure 21: Global Endosurgery Devices Market Attractiveness, by Application, 2019–2027
Figure 22: Global Endosurgery Devices Market Value Share, by End-user, 2018 and 2027
Figure 23: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Hospitals & Ambulatory Surgical Centers, 2017–2027
Figure 24: Global Endosurgery Devices Market Value (US$ Mn) and Y-o-Y Growth, by Specialty Clinics, 2017–2027
Figure 25: Global Endosurgery Devices Market Attractiveness, by End-user, 2019–2027
Figure 26: Global Endosurgery Devices Market Value Share, by Region, 2018 and 2027
Figure 27: Global Endosurgery Devices Market Attractiveness, by Region, 2019–2027
Figure 28: North America Endosurgery Devices Market Value (US$ Mn) Forecast and Y-o-Y Growth (%) Projection, 2017–2027
Figure 29: North America Endosurgery Devices Market Value Share (%), by Country, 2018 and 2027
Figure 30: North America Endosurgery Devices Market Attractiveness, by Country, 2019–2027
Figure 31: North America Endosurgery Devices Market Value Share, by Product, 2018 and 2027
Figure 32: North America Endosurgery Devices Market Attractiveness, by Product, 2019–2027
Figure 33: North America Endosurgery Devices Market Value Share Analysis, by Application, 2018 and 2027
Figure 34: North America Endosurgery Devices Market Attractiveness, by Application, 2019–2027
Figure 35: North America Endosurgery Devices Market Value Share, by End-user, 2018 and 2027
Figure 36: North America Endosurgery Devices Market Attractiveness, by End-user, 2019–2027
Figure 37: Europe Endosurgery Devices Market Value (US$ Mn) Forecast and Y-o-Y Growth (%) Projection, 2017–2027
Figure 38: Europe Endosurgery Devices Market Value Share (%), by Country/Sub-region, 2018 and 2027
Figure 39: Europe Endosurgery Devices Market Attractiveness, by Country/Sub-region, 2019–2027
Figure 40: Europe Endosurgery Devices Market Value Share, by Product, 2018 and 2027
Figure 41: Europe Endosurgery Devices Market Attractiveness, by Product, 2019–2027
Figure 42: Europe Endosurgery Devices Market Value Share Analysis, by Application, 2018 and 2027
Figure 43: Europe Endosurgery Devices Market Attractiveness, by Application, 2019–2027
Figure 44: Europe Endosurgery Devices Market Value Share, by End-user, 2018 and 2027
Figure 45: Europe Endosurgery Devices Market Attractiveness, by End-user, 2019–2027
Figure 46: Asia Pacific Endosurgery Devices Market Value (US$ Mn) Forecast and Y-o-Y Growth Projection (%), 2017–2027
Figure 47: Asia Pacific Endosurgery Devices Market Value Share (%), by Country/Sub-region, 2018 and 2027
Figure 48: Asia Pacific Endosurgery Devices Market Attractiveness, by Country/Sub-Region, 2019–2027
Figure 49: Asia Pacific Endosurgery Devices Market Value Share, by Product, 2018 and 2027
Figure 50: Asia Pacific Endosurgery Devices Market Attractiveness, by Product, 2019–2027
Figure 51: Asia Pacific Endosurgery Devices Market Value Share Analysis, by Application, 2018 and 2027
Figure 52: Asia Pacific Endosurgery Devices Market Attractiveness, by Application, 2019–2027
Figure 53: Asia Pacific Endosurgery Devices Market Value Share, by End-user, 2018 and 2027
Figure 54: Asia Pacific Endosurgery Devices Market Attractiveness, by End-user, 2019–2027
Figure 55: Latin America Endosurgery Devices Market Value (US$ Mn) Forecast and Y-o-Y Growth (%) Projection, 2017–2027
Figure 56: Latin America Endosurgery Devices Market Value Share (%), by Country/Sub-Region, 2018 and 2027
Figure 57: Latin America Endosurgery Devices Market Attractiveness, by Country/Sub-Region, 2019–2027
Figure 58: Latin America Endosurgery Devices Market Value Share, by Product, 2018 and 2027
Figure 59: Latin America Endosurgery Devices Market Attractiveness, by Product, 2019–2027
Figure 60: Latin America Endosurgery Devices Market Value Share Analysis, by Application, 2018 and 2027
Figure 61: Latin America Endosurgery Devices Market Attractiveness, by Application, 2019–2027
Figure 62: Latin America Endosurgery Devices Market Value Share, by End-user, 2018 and 2027
Figure 63: Latin America Endosurgery Devices Market Attractiveness, by End-user, 2019–2027
Figure 64: Middle East & Africa Endosurgery Devices Market Value (US$ Mn) Forecast and Y-o-Y Growth Projection (%), 2017–2027
Figure 65: Middle East & Africa Endosurgery Devices Market Value Share (%), by Country/Sub-Region, 2018 and 2027
Figure 66: Middle East & Africa Endosurgery Devices Market Attractiveness, by Country/Sub-Region, 2019–2027
Figure 67: Middle East & Africa Endosurgery Devices Market Value Share, by Product, 2018 and 2027
Figure 68: Middle East & Africa Endosurgery Devices Market Attractiveness, by Product, 2019–2027
Figure 69: Middle East & Africa Endosurgery Devices Market Value Share Analysis, by Application, 2018 and 2027
Figure 70: Middle East & Africa Endosurgery Devices Market Attractiveness, by Application, 2019–2027
Figure 71: Middle East & Africa Endosurgery Devices Market Value Share, by End-user, 2018 and 2027
Figure 72: Middle East & Africa Endosurgery Devices Market Attractiveness, by End-user, 2019–2027
Figure 73: Market Position Analysis, 2018, by Tier and Size of the Company