Reports
Reports
Analysts’ Viewpoint on Market Scenario
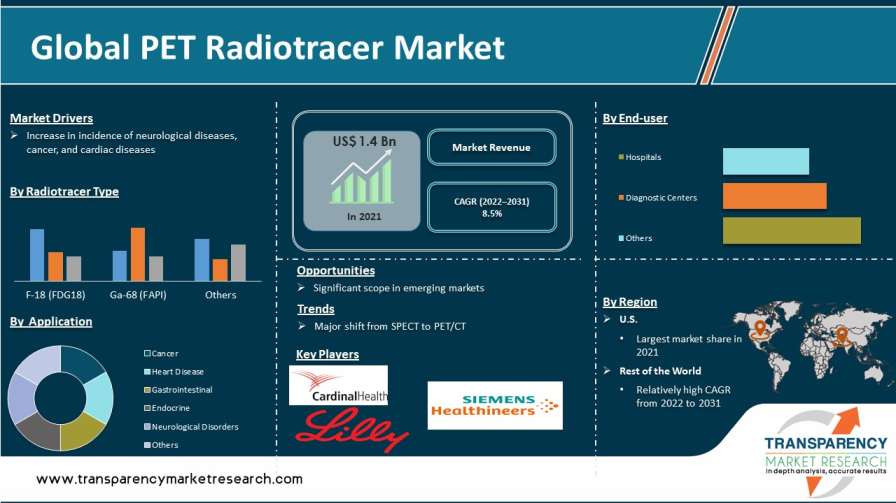
PET radiotracer, also known as PET tracer, is a positron-emitting radiopharmaceutical used in positron emission tomography (PET). Increase in incidence of neurological, cancer, and cardiac diseases; rise in capability to produce PET scan radiotracers such as Fapi radiotracer and nuclear imaging in-house; and growth in preference for PET/CT and PET scans are the key factors anticipated to drive the market size for PET radiotracers.
Key players are engaging in significant research & development activities to widen their product portfolios and thus broaden their revenue streams. For instance, on June 19, 2020, Lantheus Holdings, Inc. completed the acquisition of Progenics Pharmaceuticals, Inc., an oncology company focused on the development and commercialization of innovative targeted imaging agents.
Radiotracers used in positron emission tomography (PET) are chemical compounds comprising more than one atom replaced by a radioisotope. Each tracer consists of a positron-emitting isotope (radioactive tag) bound to an organic ligand (targeting agent).
PET radiotracers are administered intravenously. Radiotracers are used for tracking the mechanism of chemical reactions taking part in a biological system such as glucose metabolism, amino acid uptake, glucocorticoid synthesis, and other metabolic studies. Different types of radiotracers are used in PET for metabolic studies for visualization using PET imaging.
The International Atomic Energy Agency (IAEA) plays an important role in assisting the transfer of radiotracer technology to developing member states. Commonly used PET radiotracers for imaging are F 18 FDG, C-11 Methionine, F 18 fluoroethyl-l-tyrosine (FET), F 18 fluoro-dihydroxy-l-phenylalanine (F-DOPA), and C 11 Etomidate.
According to the American Nuclear Society, more than 10,000 hospitals throughout the world use radioisotopes. Nearly 90% of these are used for the diagnosis of infectious diseases, cancer, neurological disorders, and other diagnostic applications, allowing for cerebral blood flow visualization, tumor protein synthesis, neuroendocrine imaging, myocardial perfusion, and other studies.
PET is an important molecular imaging technology utilized for the assessment of neurological disorders. According to World Health Organization’s (WHO) 2021 estimates, nearly 55 million people across the globe have dementia, with Alzheimer's disease being the most common cause accounting for 60% to 70% of all dementia diagnoses.
PET scanners provide comprehensive visualization of neurological abnormalities including determination of the location of the epileptic seizures before surgery, diagnosis of movement disorders, and ability to identify and diagnose early stages of Alzheimer’s and other dementia. Diagnostic tracers for neurological indications have become a key area of interest across the world.
Large numbers of manufacturers are focusing on the introduction of tracers capable of detecting beta-amyloid plaque density in patients with cognitive impairment who are being evaluated for Alzheimer’s disease.
PET scans could be used to evaluate organs and/or tissues for the presence of disease or other conditions. PET could also be used to evaluate the function of organs such as heart and brain.
Detection of cancer and evaluation of cancer treatment are the common applications of PET, which has been recognized as a promising diagnostic tool to predict biological and physiological changes at the molecular level. Thus, PET offers a potential area for applications including stem cell research.
The PET technique allows researchers to study the normal processes in the brain (central nervous system) of normal individuals and patients with neurologic illnesses without physical/structural damage to the brain. When a region of the brain is active, it uses more fuel in the form of oxygen and sugar (glucose).
According to the American Nuclear Society, 90% of radioisotopes are used in gamma cameras or PET scan nuclear diagnostics. The remaining 10% are used in radioactive therapeutic drugs to treat diseases such as cancer and heart diseases, and gastrointestinal, endocrinal, and neurological disorders.
In terms of radiotracer type, the global market has been classified into F-18 (FDG18), Ga-68 (FAPI), and others. The F-18 (FDG18) segment dominated the global market in 2021, as FDG is the most commonly used radiotracer in clinical PET imaging. Different scientific research studies have stated that FDG PET has a sensitivity of more than 70% in detection of seizure foci as compared to other F-18 agents. This is driving the usage of FDG as a prominent PET drug tracer.
Based on radiotracer type, the global PET radiotracer market has been segregated into cancer, heart disease, gastrointestinal, endocrine, neurological disorders, and others. Most of the radioisotopes across the globe are used in oncology for diagnosis or radiation therapy. F-18 and Ga-68-based radioisotopes are primarily used in the diagnosis of cancer.
In terms of end-user, the hospitals segment dominated the global market for PET radiotracer in 2021. The trend is anticipated to continue during the forecast period. Increase in patient preference for hospitals for PET radiotracer diagnosis is expected to propel the hospitals segment during the forecast period.
Furthermore, the hospitals segment is likely to grow at a high CAGR during the forecast period owing to the increase in number of neurology, cancer, and cardiac patients; availability of advanced PET systems at low cost; and presence of trained medical personnel.
The U.S. and Europe, cumulatively, accounted for more than 95% market share for PET radiotracer in 2021. The U.S. was the dominant market in 2021. The trend is anticipated to continue during the forecast period.
Growth of the market in the U.S. can be ascribed to the increase in usage of fluorine-18 for various diseases. According to the U.S. Centers for Disease Control, an estimated 2,096,000 clinical PET scans were performed in hospital and non-hospital sites using fixed PET imaging systems or mobile PET services in 2020.
The report concludes with the company profiles section, which includes information about key players in the global PET radiotracer industry. Key players are focusing on adopting strategies such as acquisition & collaboration, geographic expansion, expansion of distribution channels, and R&D to enhance their share in the global market.
PET radiotracer companies analyzed in the report are ABX advanced biochemical compounds GmbH, Blue Earth Diagnostics, Cardinal Health, Eli Lilly and Company, GE Healthcare, IBA Radiopharma Solutions, Jubilant Pharma Limited, Lantheus Holdings, Inc., Siemens Healthineers AG, and Yantai Dongcheng Pharmaceutical Group Co., Ltd.
Each of these players has been profiled in the PET radiotracer market report based on parameters such as company overview, financial overview, business strategies, product portfolio, business segments, and recent developments.
|
Attribute |
Detail |
|
Market Size Value in 2021 |
US$ 1.4 Bn |
|
Market Forecast Value in 2031 |
More than US$ 3.2 Bn |
|
Growth Rate (CAGR) for 2022-2031 |
8.5% |
|
Forecast Period |
2022–2031 |
|
Historical Data Available for |
2017–2020 |
|
Quantitative Units |
US$ Bn for Value |
|
Market Analysis |
It includes segment analysis as well as regional level analysis. Moreover, the qualitative analysis includes PET radiotracer market drivers, restraints, opportunities, key trends, and a parent industry overview. |
|
Competition Landscape |
|
|
Format |
Electronic (PDF) + Excel |
|
Market Segmentation |
|
|
Regions Covered |
|
|
Countries Covered |
|
|
Companies Profiled |
|
|
Customization Scope |
Available upon request |
|
Pricing |
Available upon request |
The global market was valued at US$ 1.4 Bn in 2021.
The market is projected to reach more than US$ 3.2 Bn by 2031.
The global market grew at a CAGR of 3.9% from 2017 to 2021.
The global market is anticipated to grow at a CAGR of 8.5% from 2022 to 2031.
The F-18 [FDG18] segment held more than 90% share of the global PET radiotracer market in 2021.
The U.S. is expected to account for the major share of the global PET radiotracer market during the forecast period.
ABX advanced biochemical compounds GmbH, Blue Earth Diagnostics, Cardinal Health, Eli Lilly and Company, GE Healthcare, IBA Radiopharma Solutions, Jubilant Pharma Limited, Lantheus Holdings Inc., Siemens Healthineers AG, and Yantai Dongcheng Pharmaceutical Group Co., Ltd.
1. Preface
1.1. Market Definition and Scope
1.2. Market Segmentation
1.3. Key Research Objectives
1.4. Research Highlights
2. Assumptions and Research Methodology
3. Executive Summary: Global PET Radiotracer Market
4. Market Overview
4.1. Introduction
4.1.1. Radiotracer Type Definition
4.2. Overview
4.3. Market Dynamics
4.3.1. Drivers
4.3.2. Restraints
4.3.3. Opportunities
4.4. Global PET Radiotracer Market Analysis and Forecast, 2017–2031
4.4.1. Market Revenue Projections (US$ Mn)
5. Key Insights
5.1. Regulatory Scenario by Region/globally
5.2. Key Industry Events (mergers, acquisitions, partnerships, etc.)
5.3. Technological Advancements
5.4. COVID 19 Impact Analysis
6. Global PET Radiotracer Market Analysis and Forecast, by Radiotracer Type
6.1. Introduction & Definition
6.2. Key Findings / Developments
6.3. Market Value Forecast, by Radiotracer Type, 2017–2031
6.3.1. F-18 (FDG18)
6.3.2. Ga-68 (FAPI)
6.3.3. Others
6.4. Market Attractiveness Analysis, by Radiotracer Type
7. Global PET Radiotracer Market Analysis and Forecast, by Application
7.1. Introduction & Definition
7.2. Key Findings / Developments
7.3. Market Value Forecast, by Application, 2017–2031
7.3.1. Cancer
7.3.2. Heart Disease
7.3.3. Gastrointestinal
7.3.4. Endocrine
7.3.5. Neurological Disorders
7.3.6. Others
7.4. Market Attractiveness Analysis, by Application
8. Global PET Radiotracer Market Analysis and Forecast, by End-user
8.1. Introduction & Definition
8.2. Key Findings / Developments
8.3. Market Value Forecast, by End-user, 2017–2031
8.3.1. Hospitals
8.3.2. Diagnostic Centers
8.3.3. Others
8.4. Market Attractiveness Analysis, by End-user
9. Global PET Radiotracer Market Analysis and Forecast, by Country/Region
9.1. Key Findings
9.2. Market Value Forecast, by Country/Region
9.2.1. U.S.
9.2.2. Europe
9.2.3. Rest of the World
9.3. Market Attractiveness Analysis, by Country/Region
10. U.S. PET Radiotracer Market Analysis and Forecast
10.1. Introduction
10.1.1. Key Findings
10.2. Market Value Forecast, by Radiotracer Type, 2017–2031
10.2.1. F-18 (FDG18)
10.2.2. Ga-68 (FAPI)
10.2.3. Others
10.3. Market Value Forecast, by Application, 2017–2031
10.3.1. Cancer
10.3.2. Heart Disease
10.3.3. Gastrointestinal
10.3.4. Endocrine
10.3.5. Neurological Disorders
10.3.6. Others
10.4. Market Value Forecast, by End-user, 2017–2031
10.4.1. Hospitals
10.4.2. Diagnostic Centers
10.4.3. Others
10.5. Market Attractiveness Analysis
10.5.1. By Radiotracer Type
10.5.2. By Application
10.5.3. By End-user
11. Europe PET Radiotracer Market Analysis and Forecast
11.1. Introduction
11.1.1. Key Findings
11.2. Market Value Forecast, by Radiotracer Type, 2017–2031
11.2.1. F-18 (FDG18)
11.2.2. Ga-68 (FAPI)
11.2.3. Others
11.3. Market Value Forecast, by Application, 2017–2031
11.3.1. Cancer
11.3.2. Heart Disease
11.3.3. Gastrointestinal
11.3.4. Endocrine
11.3.5. Neurological Disorders
11.3.6. Others
11.4. Market Value Forecast, by End-user, 2017–2031
11.4.1. Hospitals
11.4.2. Diagnostic Centers
11.4.3. Others
11.5. Market Value Forecast, by Country/Sub-region, 2017–2031
11.5.1. Germany
11.5.2. U.K.
11.5.3. France
11.5.4. Spain
11.5.5. Italy
11.5.6. Rest of Europe
11.6. Market Attractiveness Analysis
11.6.1. By Radiotracer Type
11.6.2. By Application
11.6.3. By End-user
11.6.4. By Country/Sub-region
12. Rest of the World PET Radiotracer Market Analysis and Forecast
12.1. Introduction
12.1.1. Key Findings
12.2. Market Value Forecast, by Radiotracer Type, 2017–2031
12.2.1. F-18 (FDG18)
12.2.2. Ga-68 (FAPI)
12.2.3. Others
12.3. Market Value Forecast, by Application, 2017–2031
12.3.1. Cancer
12.3.2. Heart Disease
12.3.3. Gastrointestinal
12.3.4. Endocrine
12.3.5. Neurological Disorders
12.3.6. Others
12.4. Market Value Forecast, by End-user, 2017–2031
12.4.1. Hospitals
12.4.2. Diagnostic Centers
12.4.3. Others
12.5. Market Attractiveness Analysis
12.5.1. By Radiotracer Type
12.5.2. By Application
12.5.3. By End-user
13. Competition Landscape
13.1. Market Player – Competition Matrix (By Tier and Size of companies)
13.2. Market Share Analysis By Company (2021)
13.3. Company Profiles
13.3.1. ABX advanced biochemical compounds GmbH
13.3.1.1. Company Overview
13.3.1.2. Strategic Overview
13.3.1.3. SWOT Analysis
13.3.2. Blue Earth Diagnostics
13.3.2.1. Company Overview
13.3.2.2. Financial Overview
13.3.2.3. Strategic Overview
13.3.2.4. SWOT Analysis
13.3.3. Cardinal Health
13.3.3.1. Company Overview
13.3.3.2. Financial Overview
13.3.3.3. Strategic Overview
13.3.3.4. SWOT Analysis
13.3.4. Eli Lilly and Company
13.3.4.1. Company Overview
13.3.4.2. Financial Overview
13.3.4.3. Strategic Overview
13.3.4.4. SWOT Analysis
13.3.5. GE Healthcare
13.3.5.1. Company Overview
13.3.5.2. Financial Overview
13.3.5.3. Strategic Overview
13.3.5.4. SWOT Analysis
13.3.6. IBA Radiopharma Solutions
13.3.6.1. Company Overview
13.3.6.2. Financial Overview
13.3.6.3. Strategic Overview
13.3.6.4. SWOT Analysis
13.3.7. Jubilant Pharma Limited
13.3.7.1. Company Overview
13.3.7.2. Financial Overview
13.3.7.3. Strategic Overview
13.3.7.4. SWOT Analysis
13.3.8. Lantheus Holdings, Inc.
13.3.8.1. Company Overview
13.3.8.2. Financial Overview
13.3.8.3. Strategic Overview
13.3.8.4. SWOT Analysis
13.3.9. Siemens Healthineers AG
13.3.9.1. Company Overview
13.3.9.2. Financial Overview
13.3.9.3. Strategic Overview
13.3.9.4. SWOT Analysis
13.3.10. Yantai Dongcheng Pharmaceutical Group Co., Ltd.
13.3.10.1. Company Overview
13.3.10.2. Financial Overview
13.3.10.3. Strategic Overview
13.3.10.4. SWOT Analysis
List of Tables
Table 01: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Radiotracer Type, 2017–2031
Table 02: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Application, 2017–2031
Table 03: Global PET Radiotracer Market Value (US$ Mn) Forecast, by End-user, 2017–2031
Table 04: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Country/Region, 2017–2031
Table 05: U.S. PET Radiotracer Market Value (US$ Mn) Forecast, by Radiotracer Type, 2017–2031
Table 06: U.S. PET Radiotracer Market Value (US$ Mn) Forecast, by Application, 2017–2031
Table 07: U.S. PET Radiotracer Market Value (US$ Mn) Forecast, by End-user, 2017–2031
Table 08: Europe PET Radiotracer Market Value (US$ Mn) Forecast, by Radiotracer Type, 2017–2031
Table 09: Europe PET Radiotracer Market Value (US$ Mn) Forecast, by Application, 2017–2031
Table 10: Europe PET Radiotracer Market Value (US$ Mn) Forecast, by End-user, 2017–2031
Table 11: Europe PET Radiotracer Market Value (US$ Mn) Forecast, by Country/Sub-region, 2017–2031
Table 12: Rest of the World PET Radiotracer Market Value (US$ Mn) Forecast, by Radiotracer Type, 2017–2031
Table 13: Rest of the World PET Radiotracer Market Value (US$ Mn) Forecast, by Application, 2017–2031
Table 14: Rest of the World PET Radiotracer Market Value (US$ Mn) Forecast, by End-user, 2017–2031
List of Figures
Figure 01: Global PET Radiotracer Market Value (US$ Mn) Forecast and Y-o-Y Growth (%) Projection, 2017–2031
Figure 02: Global PET Radiotracer Market Value Share Analysis, by Radiotracer Type, 2021 and 2031
Figure 03: Global PET Radiotracer Market Attractiveness Analysis, by Radiotracer Type, 2022–2031
Figure 04: Global PET Radiotracer Market Value (US$ Mn) Forecast, by F-18 (FDG18), 2017–2031
Figure 05: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Ga-68 (FAPI), 2017–2031
Figure 06: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Others, 2017–2031
Figure 07: Global PET Radiotracer Market Value Share Analysis, by Application, 2021 and 2031
Figure 08: Global PET Radiotracer Market Attractiveness Analysis, by Application, 2022–2031
Figure 09: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Cancer, 2017–2031
Figure 10: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Heart Disease, 2017–2031
Figure 11: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Gastrointestinal, 2017–2031
Figure 12: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Endocrine, 2017–2031
Figure 13: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Neurological Disorders, 2017–2031
Figure 14: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Others, 2017–2031
Figure 15: Global PET Radiotracer Market Value Share Analysis, by End-user, 2021 and 2031
Figure 16: Global PET Radiotracer Market Attractiveness Analysis, by End-user, 2022–2031
Figure 17: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Hospitals, 2017–2031
Figure 18: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Diagnostic Centers, 2017–2031
Figure 19: Global PET Radiotracer Market Value (US$ Mn) Forecast, by Others, 2017–2031
Figure 20: Global PET Radiotracer Market Value Share Analysis, by Country/Region, 2021 and 2031
Figure 21: Global PET Radiotracer Market Attractiveness Analysis, by Country/Region, 2022–2031
Figure 22: U.S. PET Radiotracer Market Value (US$ Mn) Forecast, 2017–2031
Figure 23: U.S. PET Radiotracer Market Value Share Analysis, by Radiotracer Type, 2021 and 2031
Figure 24: U.S. PET Radiotracer Market Attractiveness Analysis, by Radiotracer Type, 2022–2031
Figure 25: U.S. PET Radiotracer Market Value Share Analysis, by Application, 2021 and 2031
Figure 26: U.S. PET Radiotracer Market Attractiveness Analysis, by Application, 2022–2031
Figure 27: U.S. PET Radiotracer Market Value Share Analysis, by End-user, 2021 and 2031
Figure 28: U.S. PET Radiotracer Market Attractiveness Analysis, by End-user, 2022–2031
Figure 29: Europe PET Radiotracer Market Value (US$ Mn) Forecast, 2017–2031
Figure 30: Europe PET Radiotracer Market Value Share Analysis, by Radiotracer Type, 2021 and 2031
Figure 31: Europe PET Radiotracer Market Attractiveness Analysis, by Radiotracer Type, 2022–2031
Figure 32: Europe PET Radiotracer Market Value Share Analysis, by Application, 2021 and 2031
Figure 33: Europe PET Radiotracer Market Attractiveness Analysis, by Application, 2022–2031
Figure 34: Europe PET Radiotracer Market Value Share Analysis, by End-user, 2021 and 2031
Figure 35: Europe PET Radiotracer Market Attractiveness Analysis, by End-user, 2022–2031
Figure 36: Europe PET Radiotracer Market Value Share Analysis, by Country/Sub-region, 2021 and 2031
Figure 37: Europe PET Radiotracer Market Attractiveness Analysis, by Country/Sub-region, 2022–2031
Figure 38: Rest of the World PET Radiotracer Market Value (US$ Mn) Forecast, 2017–2031
Figure 39: Rest of the World PET Radiotracer Market Value Share Analysis, by Radiotracer Type, 2021 and 2031
Figure 40: Rest of the World PET Radiotracer Market Attractiveness Analysis, by Radiotracer Type, 2022–2031
Figure 41: Rest of the World PET Radiotracer Market Value Share Analysis, by Application, 2021 and 2031
Figure 42: Rest of the World PET Radiotracer Market Attractiveness Analysis, by Application, 2022–2031
Figure 43: Rest of the World PET Radiotracer Market Value Share Analysis, by End-user, 2021 and 2031
Figure 44: Rest of the World PET Radiotracer Market Attractiveness Analysis, by End-user, 2022–2031