Reports
Reports
The ophthalmic drug market is currently witnessing robust growth because of increased prevalence of eye diseases, advancements in pharmaceutical technology, and enhanced global awareness of the importance of eye care. One of the major reasons for this market expansion is rise in the prevalence of eye disorders.
Glaucoma, diabetic retinopathy, and cataracts are mainly being driven by aging population and lifestyle changes. As the global population is gradually moving toward older demographics, there is an increased need for treatment modalities to treat these conditions effectively.
.webp)
Moreover, the speed at which drug delivery systems and drug formulations are improving, along with the ongoing investigations into advanced treatments like gene therapy and controlled-release drug delivery systems widening the treatment options window for patients. Not only do these advances improve the efficacy of drugs, but also the compliance of patients to follow the treatment regimen.
Furthermore, increased investment by the pharmaceutical industry into research and development activities is also resulting in an increased manufacturing of new and innovative ophthalmic pharmaceuticals. The business climate also increases with partnerships involving biopharmaceutical companies and university in an effort to develop new therapeutic modalities.
The ophthalmic drugs industry is also experiencing new developments through telemedicine and online health platforms with remote diagnosis and monitoring, thereby allowing for earlier treatments and more effective management of disease.
Additionally, one of the most critical driving forces is the increasing recognition of the need for eye care due to campaigns and publicity programmes that have raised public awareness. More evolved consumers with respect to threats to their eye care drive preventive treatment and care, which increases the demand for ophthalmic products.
Additionally, an increase in penetration of ophthalmic drugs in traditional as well as online pharmacies provides enhanced patient access and thus drives market expansion. The rise in threat perception toward eye health, along with higher defensive and precautionary measures taken by customers, leads to higher demand for ophthalmic products. In addition, the higher availability of ophthalmic drugs through retail pharmacies as well as online pharmacies raises patient accessibility, thereby driving market growth.
Ophthalmic medications are unique drugs developed for the diagnosis, treatment, and prevention of a multitude of conditions and diseases related to the eye. They are formulated and designed to be administered to the eye directly to limit side-effects and allow for action by sufficiently applying drug to the site of action. The uses of ophthalmic medications are diverse and come in a variety of classes of therapy.
Ophthalmic medications come in many formulations and can be classified in many different ways, including anti-inflammatory medications, antibiotics, antifungals, antivirals, and local anesthetics. For example, corticosteroids are used to diminish inflammation following surgery or trauma to the eye, while antibiotics are necessary for treating awful bacterial infection such as conjunctivitis. Prostaglandin analogues and beta-blockers also play an important role in treating intraocular pressure in patients with glaucoma and hence avoiding blindness.
Recent advances in drug design have allowed the creation of new drug delivery systems like sustained-release implants and nanotechnology products, which are more effective and of longer therapeutic duration. These ease the dosing regimens and therefore enhance patient compliance with therapy.
Additionally, ophthalmic medication availability can be categorized into two major groups: prescription medication and over-the-counter drugs. Prescription ophthalmic medication is typically applied in more severe eye problems that need to be clinically diagnosed and treated. These involve glaucoma medication, infection, inflammatory medication, and some chronic eye diseases.
Prescription medication generally carries strong active ingredients that must be strictly monitored by physicians to be safe and effective. For example, drugs like glaucoma prostaglandin analogs or corticosteroids to reduce inflammation require cautious dosing and monitoring to avoid any side effects.
In contrast, over-the-counter ophthalmic medication is readily available to the ordinary consumer without prescription. These are typically applied for the management of milder eye conditions such as dry eyes, allergies, and minor irritations. Best-selling over-the-counter ophthalmic medication includes artificial tears, antihistamine drops, and redness reducers. Their availability allows patients to manage minor symptoms simply and effectively without going to the physician.
| Attribute | Detail |
|---|---|
| Ophthalmic Drugs Market Drivers |
|
The increasing prevalence of eye diseases is a key driver to the market for ophthalmic pharmaceuticals, a significant public health issue and emerging business opportunity for the pharmaceutical sector. With rise in aging population worldwide, the incidences of most eye diseases have increased, thereby posing a pressing requirement for effective treatment. Age-related macular degeneration (AMD), glaucoma, diabetic retinopathy, and cataracts are among the most common disorders affecting vision, especially in older adults.
According to the World Health Organization, it is estimated that 2.2 billion individuals are currently living with near or distance visual impairment. Of these, at least 1 billion cases could have been avoided or remain to be treated. Blindness and vision impairment also have a colossal economic impact at the international level, and the overall yearly cost of productivity has been estimated to be US$ 411.0 Bn.
One of the most causative drivers to increasing cases of eye diseases is the ongoing trend of an aging demographic. Since there is improved life expectancy, more individuals live with ocular ailments. For example, conditions like glaucoma, which, if left untreated, lead to irreversible blindness, are prevalent among elderly individuals. Also, diabetic retinopathy is one of the causes of visual disability in the working age population, and hence management prefers to keep it under control.
Additionally, lifestyle variables like extended screen use, bad eating habits, and physical inactivity have contributed to the onset of eye diseases. The technology era has added a further disease burden in the form of digital eye fatigue, which occurs with symptoms of dryness, blurred vision, and irritation. Increasingly, this disorder is being acknowledged, and this has led to demand for over-the-counter drugs like lubricating eye drops, which are the prime ingredients of ophthalmic drugs market.
The increasing need for telemedicine and digital health solutions is a paradigm shift in the ophthalmic drugs market, which is transforming delivery paradigms of eye care and patient treatment. Telemedicine has witnessed explosive growth over the last few years, and particularly due to the COVID-19 pandemic requiring remote consultations and treatments. The technology has enabled the patients to be diagnosed and treated for their eye ailments in time without having to report alone, thereby enhancing the ophthalmic service accessibility.
The most significant benefit of telemedicine in ophthalmology is that it can extend to underserved populations, such as rural or remote communities where access to specialty eye care may be lacking. Through digital technology healthcare professionals can perform virtual visits, thereby allowing patients to report symptoms, receive preliminary diagnoses, and obtain prescriptions for eye medications from home. This increased access not only allows for earlier diagnosis and treatment of eye disease but also prompts patients who may otherwise be reluctant to report for treatment due to logistical reasons to report for timely treatment.
Electronic health platforms like wearable devices and mobile health applications further advance this setting by providing patients with the authority of proactiveness in their own eye care. For example, mobile applications may remind patients to comply with medication, allow tracking of symptoms, and even remotely monitor intraocular pressure in glaucoma patients.
All these equipment encourage increase patient compliance and cooperation that are vital to the effective management of chronic eye disease.
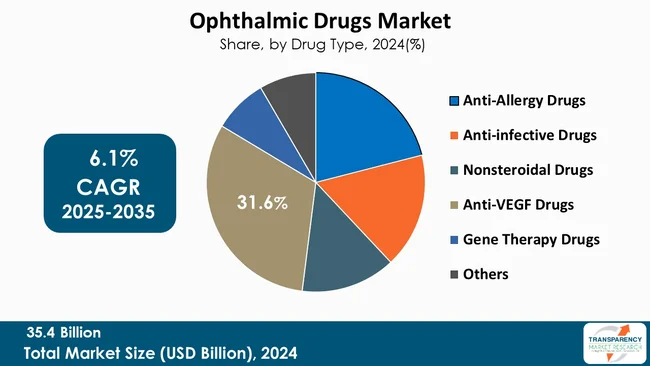
Anti-VEGF (Vascular Endothelial Growth Factor) Drugs segment leads the global ophthalmic drugs market. This can be attributed to the use of such medications in treating prevalent eye conditions such as age-related macular degeneration (AMD), diabetic retinopathy, and retinal vein occlusion, which are the leading causes of vision impairment globally.
Pegaptanib (Macugen), an aptamer targeting VEGF-A165, was the first intravitreal drug approved for treating wet AMD. In recent years, VEGF inhibitors such as bevacizumab (Avastin), ranibizumab (Lucentis), and aflibercept (Eylea) have been commonly used in clinical practice.
| Attribute | Detail |
|---|---|
| Leading Region | North America |
As per the latest ophthalmic drugs market analysis, North America dominated the market in 2024. The region is equipped with a well-established healthcare system, advanced diagnostic and treatment facilities. The center facilitates timely access to eye care services, encouraging the use of innovative treatments and medicines.
Further, high incidence of eye diseases like age-related macular degeneration, diabetic retinopathy, and glaucoma contributes to the demand for ophthalmic drugs. Demand is further fueled by the aging U.S. and Canadian population since these conditions are more prevalent in older people.
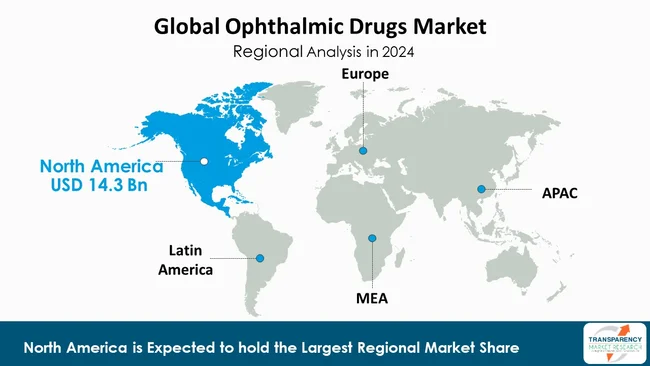
North America houses huge pharmaceutical R&D, with the majority of top companies and R&D facilities spending heavily on ophthalmic drug developments. The high concentration of R&D allows new treatments to enter the market, advancing the treatment for individuals.
Some of the important initiatives undertaken by companies in the ophthalmic drugs market are significant investment in R&D for the development of anti-VEGF therapy, gene therapy, and new drug delivery systems for enhanced efficacy and patient compliance. The major pharmaceutical companies are conducting high-scale clinical trials to evaluate the safety and efficacy of drugs.
Alcon Inc., Novartis AG, Bausch + Lomb., Merck & Co., Inc., Regeneron Pharmaceuticals Inc., Coherus BioSciences, Inc., Pfizer Inc., AbbVie, RIBOMIC, Santen Pharmaceutical Co., Ltd., Cipla Limited, Lupin Pharmaceuticals, Inc., Ocular Therapeutix, Inc., Novaliq GmbH, ZEXUS PHARMA, and Grevis Pharmaceuticals Pvt. Ltd. are some of the leading players operating in the global ophthalmic drugs market.
Each of these players has been profiled in the ophthalmic drugs market research report based on parameters such as company overview, financial overview, business strategies, product portfolio, business segments, and recent developments.
| Attribute | Detail |
|---|---|
| Size in 2024 | US$ 35.4 Bn |
| Forecast Value in 2035 | US$ 67.9 Bn |
| CAGR | 6.1% |
| Forecast Period | 2025-2035 |
| Historical Data Available for | 2020-2023 |
| Quantitative Units | US$ Bn |
| Biotechnology Market Analysis | It includes segment analysis as well as regional level analysis. Moreover, qualitative analysis includes drivers, restraints, opportunities, key trends, value chain analysis, and key trend analysis. |
| Competition Landscape |
|
| Format | Electronic (PDF) + Excel |
| Segmentation |
|
| Regions Covered |
|
| Countries Covered |
|
| Companies Profiled |
|
| Customization Scope | Available upon request |
| Pricing | Available upon request |
The global ophthalmic drugs market was valued at US$ 35.4 Bn in 2024.
The global ophthalmic drugs market is projected to reach more than US$ 67.9 Bn by the end of 2035.
Increasing prevalence of eye disorders, growing global geriatric population, advancements in drug formulations, delivery systems, and surgical technologies, and increased awareness of eye health are the factors driving the expansion of ophthalmic drugs market.
The CAGR is anticipated to be 6.1% from 2025 to 2035.
Alcon Inc., Novartis AG, Bausch + Lomb., Merck & Co., Inc., Regeneron Pharmaceuticals Inc., Coherus BioSciences, Inc., Pfizer Inc., AbbVie, RIBOMIC, Santen Pharmaceutical Co., Ltd., Cipla Limited, Lupin Pharmaceuticals, Inc., Ocular Therapeutix, Inc., Novaliq GmbH, ZEXUS PHARMA, and Grevis Pharmaceuticals Pvt. Ltd.
1. Preface
1.1. Market Definition and Scope
1.2. Market Segmentation
1.3. Key Research Objectives
1.4. Research Highlights
2. Assumptions and Research Methodology
3. Executive Summary: Global Ophthalmic Drugs Market
4. Market Overview
4.1. Introduction
4.1.1. Segment Definition
4.2. Overview
4.3. Market Dynamics
4.3.1. Drivers
4.3.2. Restraints
4.3.3. Opportunities
4.4. Global Ophthalmic Drugs Market Analysis and Forecasts, 2020 to 2035
4.4.1. Market Revenue Projections (US$ Bn)
5. Key Insights
5.1. Healthcare Expenditure across Key Regions / Countries
5.2. Recent Advancements in Ophthalmic Medications
5.3. Epidemiology of Ophthalmic Disorders across Key Regions / Countries
5.4. Pricing Trends for Ophthalmic Drugs
5.5. Regulatory Scenario across Key Regions / Countries
5.6. PORTER’s Five Forces Analysis
5.7. PESTEL Analysis
5.8. Value Chain Analysis
5.9. Key Purchase Metrics for End-users
5.10. Go-to-Market Strategy for New Market Entrants
5.11. Key Industry Events (Partnerships, Collaborations, Product approvals, mergers & acquisitions)
5.12. Benchmarking of Key Products Offered by the Leading Competitors
6. Global Ophthalmic Drugs Market Analysis and Forecasts, By Drug Type
6.1. Introduction & Definition
6.2. Key Findings / Developments
6.3. Market Value Forecast By Drug Type, 2020 to 2035
6.3.1. Anti-Allergy Drugs
6.3.2. Anti-infective Drugs
6.3.3. Nonsteroidal Drugs
6.3.4. Anti-VEGF Drugs
6.3.4.1. Aptamer
6.3.4.2. Antibodies (mAb & Bispecific Antibody)
6.3.4.3. Fusion Protein
6.3.5. Gene Therapy Drugs
6.3.6. Others
6.4. Market Attractiveness By Drug Type
7. Global Ophthalmic Drugs Market Analysis and Forecasts, By Route of Administration
7.1. Introduction & Definition
7.2. Key Findings / Developments
7.3. Market Value Forecast By Route of Administration, 2020 to 2035
7.3.1. Topical
7.3.2. Intraocular
7.3.3. Others
7.4. Market Attractiveness By Route of Administration
8. Global Ophthalmic Drugs Market Analysis and Forecasts, By Dosage Form
8.1. Introduction & Definition
8.2. Key Findings / Developments
8.3. Market Value Forecast By Dosage Form, 2020 to 2035
8.3.1. Gel
8.3.2. Drop
8.3.3. Ointment
8.3.4. Suspension & Solutions
8.3.5. Others
8.4. Market Attractiveness By Dosage Form
9. Global Ophthalmic Drugs Market Analysis and Forecasts, By Product Type
9.1. Introduction & Definition
9.2. Key Findings / Developments
9.3. Market Value Forecast By Product Type, 2020 to 2035
9.3.1. Prescription Drugs
9.3.2. Over-the-Counter
9.4. Market Attractiveness By Product Type
10. Global Ophthalmic Drugs Market Analysis and Forecasts, By Indication
10.1. Introduction & Definition
10.2. Key Findings / Developments
10.3. Market Value Forecast By Indication, 2020 to 2035
10.3.1. Glaucoma
10.3.2. Dry Eye Disease
10.3.3. Age-Related Macular Degeneration
10.3.4. Diabetic Retinopathy
10.3.5. Pink Eye
10.3.6. Cataracts
10.3.7. Retinal Detachment
10.3.8. Others
10.4. Market Attractiveness By Indication
11. Global Ophthalmic Drugs Market Analysis and Forecasts, By Distribution Channel
11.1. Introduction & Definition
11.2. Key Findings / Developments
11.3. Market Value Forecast By Distribution Channel, 2020 to 2035
11.3.1. Hospital Pharmacies
11.3.2. Retail Pharmacies
11.3.3. Online Pharmacies
11.4. Market Attractiveness By Distribution Channel
12. Global Ophthalmic Drugs Market Analysis and Forecasts, By Region
12.1. Key Findings
12.2. Market Value Forecast By Region
12.2.1. North America
12.2.2. Europe
12.2.3. Asia Pacific
12.2.4. Latin America
12.2.5. Middle East & Africa
12.3. Market Attractiveness By Region
13. North America Ophthalmic Drugs Market Analysis and Forecast
13.1. Introduction
13.1.1. Key Findings
13.2. Market Value Forecast By Drug Type, 2020 to 2035
13.2.1. Anti-Allergy Drugs
13.2.2. Anti-infective Drugs
13.2.3. Nonsteroidal Drugs
13.2.4. Anti-VEGF Drugs
13.2.4.1. Aptamer
13.2.4.2. Antibodies (mAb & Bispecific Antibody)
13.2.4.3. Fusion Protein
13.2.5. Gene Therapy Drugs
13.2.6. Others
13.3. Market Value Forecast By Route of Administration, 2020 to 2035
13.3.1. Topical
13.3.2. Intraocular
13.3.3. Others
13.4. Market Value Forecast By Dosage Form, 2020 to 2035
13.4.1. Gel
13.4.2. Drop
13.4.3. Ointment
13.4.4. Suspension & Solutions
13.4.5. Others
13.5. Market Value Forecast By Product Type, 2020 to 2035
13.5.1. Prescription Drugs
13.5.2. Over-the-Counter
13.6. Market Value Forecast By Indication, 2020 to 2035
13.6.1. Glaucoma
13.6.2. Dry Eye Disease
13.6.3. Age-Related Macular Degeneration
13.6.4. Diabetic Retinopathy
13.6.5. Pink Eye
13.6.6. Cataracts
13.6.7. Retinal Detachment
13.6.8. Others
13.7. Market Value Forecast By Distribution Channel, 2020 to 2035
13.7.1. Hospital Pharmacies
13.7.2. Retail Pharmacies
13.7.3. Online Pharmacies
13.8. Market Value Forecast By Country , 2020 to 2035
13.8.1. U.S.
13.8.2. Canada
13.9. Market Attractiveness Analysis
13.9.1. By Drug Type
13.9.2. By Route of Administration
13.9.3. By Dosage Form
13.9.4. By Product Type
13.9.5. By Indication
13.9.6. By Distribution Channel
13.9.7. By Country
14. Europe Ophthalmic Drugs Market Analysis and Forecast
14.1. Introduction
14.1.1. Key Findings
14.2. Market Value Forecast By Drug Type, 2020 to 2035
14.2.1. Anti-Allergy Drugs
14.2.2. Anti-infective Drugs
14.2.3. Nonsteroidal Drugs
14.2.4. Anti-VEGF Drugs
14.2.4.1. Aptamer
14.2.4.2. Antibodies (mAb & Bispecific Antibody)
14.2.4.3. Fusion Protein
14.2.5. Gene Therapy Drugs
14.2.6. Others
14.3. Market Value Forecast By Route of Administration, 2020 to 2035
14.3.1. Topical
14.3.2. Intraocular
14.3.3. Others
14.4. Market Value Forecast By Dosage Form, 2020 to 2035
14.4.1. Gel
14.4.2. Drop
14.4.3. Ointment
14.4.4. Suspension & Solutions
14.4.5. Others
14.5. Market Value Forecast By Product Type, 2020 to 2035
14.5.1. Prescription Drugs
14.5.2. Over-the-Counter
14.6. Market Value Forecast By Indication, 2020 to 2035
14.6.1. Glaucoma
14.6.2. Dry Eye Disease
14.6.3. Age-Related Macular Degeneration
14.6.4. Diabetic Retinopathy
14.6.5. Pink Eye
14.6.6. Cataracts
14.6.7. Retinal Detachment
14.6.8. Others
14.7. Market Value Forecast By Distribution Channel, 2020 to 2035
14.7.1. Hospital Pharmacies
14.7.2. Retail Pharmacies
14.7.3. Online Pharmacies
14.8. Market Value Forecast By Country / Sub-region , 2020 to 2035
14.8.1. Germany
14.8.2. UK
14.8.3. France
14.8.4. Italy
14.8.5. Spain
14.8.6. Switzerland
14.8.7. The Netherlands
14.8.8. Rest of Europe
14.9. Market Attractiveness Analysis
14.9.1. By Drug Type
14.9.2. By Route of Administration
14.9.3. By Dosage Form
14.9.4. By Product Type
14.9.5. By Indication
14.9.6. By Distribution Channel
14.9.7. By Country / Sub-region
15. Asia Pacific Ophthalmic Drugs Market Analysis and Forecast
15.1. Introduction
15.1.1. Key Findings
15.2. Market Value Forecast By Drug Type, 2020 to 2035
15.2.1. Anti-Allergy Drugs
15.2.2. Anti-infective Drugs
15.2.3. Nonsteroidal Drugs
15.2.4. Anti-VEGF Drugs
15.2.4.1. Aptamer
15.2.4.2. Antibodies (mAb & Bispecific Antibody)
15.2.4.3. Fusion Protein
15.2.5. Gene Therapy Drugs
15.2.6. Others
15.3. Market Value Forecast By Route of Administration, 2020 to 2035
15.3.1. Topical
15.3.2. Intraocular
15.3.3. Others
15.4. Market Value Forecast By Dosage Form, 2020 to 2035
15.4.1. Gel
15.4.2. Drop
15.4.3. Ointment
15.4.4. Suspension & Solutions
15.4.5. Others
15.5. Market Value Forecast By Product Type, 2020 to 2035
15.5.1. Prescription Drugs
15.5.2. Over-the-Counter
15.6. Market Value Forecast By Indication, 2020 to 2035
15.6.1. Glaucoma
15.6.2. Dry Eye Disease
15.6.3. Age-Related Macular Degeneration
15.6.4. Diabetic Retinopathy
15.6.5. Pink Eye
15.6.6. Cataracts
15.6.7. Retinal Detachment
15.6.8. Others
15.7. Market Value Forecast By Distribution Channel, 2020 to 2035
15.7.1. Hospital Pharmacies
15.7.2. Retail Pharmacies
15.7.3. Online Pharmacies
15.8. Market Value Forecast By Country / Sub-region , 2020 to 2035
15.8.1. China
15.8.2. India
15.8.3. Japan
15.8.4. South Korea
15.8.5. Australia & New Zealand
15.8.6. Rest of Asia Pacific
15.9. Market Attractiveness Analysis
15.9.1. By Drug Type
15.9.2. By Route of Administration
15.9.3. By Dosage Form
15.9.4. By Product Type
15.9.5. By Indication
15.9.6. By Distribution Channel
15.9.7. By Country / Sub-region
16. Latin America Ophthalmic Drugs Market Analysis and Forecast
16.1. Introduction
16.1.1. Key Findings
16.2. Market Value Forecast By Drug Type, 2020 to 2035
16.2.1. Anti-Allergy Drugs
16.2.2. Anti-infective Drugs
16.2.3. Nonsteroidal Drugs
16.2.4. Anti-VEGF Drugs
16.2.4.1. Aptamer
16.2.4.2. Antibodies (mAb & Bispecific Antibody)
16.2.4.3. Fusion Protein
16.2.5. Gene Therapy Drugs
16.2.6. Others
16.3. Market Value Forecast By Route of Administration, 2020 to 2035
16.3.1. Topical
16.3.2. Intraocular
16.3.3. Others
16.4. Market Value Forecast By Dosage Form, 2020 to 2035
16.4.1. Gel
16.4.2. Drop
16.4.3. Ointment
16.4.4. Suspension & Solutions
16.4.5. Others
16.5. Market Value Forecast By Product Type, 2020 to 2035
16.5.1. Prescription Drugs
16.5.2. Over-the-Counter
16.6. Market Value Forecast By Indication, 2020 to 2035
16.6.1. Glaucoma
16.6.2. Dry Eye Disease
16.6.3. Age-Related Macular Degeneration
16.6.4. Diabetic Retinopathy
16.6.5. Pink Eye
16.6.6. Cataracts
16.6.7. Retinal Detachment
16.6.8. Others
16.7. Market Value Forecast By Distribution Channel, 2020 to 2035
16.7.1. Hospital Pharmacies
16.7.2. Retail Pharmacies
16.7.3. Online Pharmacies
16.8. Market Value Forecast By Country / Sub-region , 2020 to 2035
16.8.1. Brazil
16.8.2. Mexico
16.8.3. Argentina
16.8.4. Rest of Latin America
16.9. Market Attractiveness Analysis
16.9.1. By Drug Type
16.9.2. By Route of Administration
16.9.3. By Dosage Form
16.9.4. By Product Type
16.9.5. By Indication
16.9.6. By Distribution Channel
16.9.7. By Country / Sub-region
17. Middle East & Africa Ophthalmic Drugs Market Analysis and Forecast
17.1. Introduction
17.1.1. Key Findings
17.2. Market Value Forecast By Drug Type, 2020 to 2035
17.2.1. Anti-Allergy Drugs
17.2.2. Anti-infective Drugs
17.2.3. Nonsteroidal Drugs
17.2.4. Anti-VEGF Drugs
17.2.4.1. Aptamer
17.2.4.2. Antibodies (mAb & Bispecific Antibody)
17.2.4.3. Fusion Protein
17.2.5. Gene Therapy Drugs
17.2.6. Others
17.3. Market Value Forecast By Route of Administration, 2020 to 2035
17.3.1. Topical
17.3.2. Intraocular
17.3.3. Others
17.4. Market Value Forecast By Dosage Form, 2020 to 2035
17.4.1. Gel
17.4.2. Drop
17.4.3. Ointment
17.4.4. Suspension & Solutions
17.4.5. Others
17.5. Market Value Forecast By Product Type, 2020 to 2035
17.5.1. Prescription Drugs
17.5.2. Over-the-Counter
17.6. Market Value Forecast By Indication, 2020 to 2035
17.6.1. Glaucoma
17.6.2. Dry Eye Disease
17.6.3. Age-Related Macular Degeneration
17.6.4. Diabetic Retinopathy
17.6.5. Pink Eye
17.6.6. Cataracts
17.6.7. Retinal Detachment
17.6.8. Others
17.7. Market Value Forecast By Distribution Channel, 2020 to 2035
17.7.1. Hospital Pharmacies
17.7.2. Retail Pharmacies
17.7.3. Online Pharmacies
17.8. Market Value Forecast By Country / Sub-region , 2020 to 2035
17.8.1. GCC Countries
17.8.2. South Africa
17.8.3. Rest of Middle East & Africa
17.9. Market Attractiveness Analysis
17.9.1. By Drug Type
17.9.2. By Route of Administration
17.9.3. By Dosage Form
17.9.4. By Product Type
17.9.5. By Indication
17.9.6. By Distribution Channel
17.9.7. By Country / Sub-region
18. Competition Landscape
18.1. Market Player – Competition Matrix (By Tier and Size of companies)
18.2. Market Share Analysis By Company (2024)
18.3. Company Profiles
18.3.1. Alcon Inc.
18.3.1.1. Company Overview
18.3.1.2. Financial Overview
18.3.1.3. Product Portfolio
18.3.1.4. Business Strategies
18.3.1.5. Recent Developments
18.3.2. Novartis AG
18.3.2.1. Company Overview
18.3.2.2. Financial Overview
18.3.2.3. Product Portfolio
18.3.2.4. Business Strategies
18.3.2.5. Recent Developments
18.3.3. Bausch + Lomb.
18.3.3.1. Company Overview
18.3.3.2. Financial Overview
18.3.3.3. Product Portfolio
18.3.3.4. Business Strategies
18.3.3.5. Recent Developments
18.3.4. Merck & Co., Inc.
18.3.4.1. Company Overview
18.3.4.2. Financial Overview
18.3.4.3. Product Portfolio
18.3.4.4. Business Strategies
18.3.4.5. Recent Developments
18.3.5. Regeneron Pharmaceuticals Inc.
18.3.5.1. Company Overview
18.3.5.2. Financial Overview
18.3.5.3. Product Portfolio
18.3.5.4. Business Strategies
18.3.5.5. Recent Developments
18.3.6. Coherus BioSciences, Inc.
18.3.6.1. Company Overview
18.3.6.2. Financial Overview
18.3.6.3. Product Portfolio
18.3.6.4. Business Strategies
18.3.6.5. Recent Developments
18.3.7. Pfizer Inc.
18.3.7.1. Company Overview
18.3.7.2. Financial Overview
18.3.7.3. Product Portfolio
18.3.7.4. Business Strategies
18.3.7.5. Recent Developments
18.3.8. AbbVie
18.3.8.1. Company Overview
18.3.8.2. Financial Overview
18.3.8.3. Product Portfolio
18.3.8.4. Business Strategies
18.3.8.5. Recent Developments
18.3.9. RIBOMIC
18.3.9.1. Company Overview
18.3.9.2. Financial Overview
18.3.9.3. Product Portfolio
18.3.9.4. Business Strategies
18.3.9.5. Recent Developments
18.3.10. Santen Pharmaceutical Co., Ltd.
18.3.10.1. Company Overview
18.3.10.2. Financial Overview
18.3.10.3. Product Portfolio
18.3.10.4. Business Strategies
18.3.10.5. Recent Developments
18.3.11. Cipla Limited
18.3.11.1. Company Overview
18.3.11.2. Financial Overview
18.3.11.3. Product Portfolio
18.3.11.4. Business Strategies
18.3.11.5. Recent Developments
18.3.12. Lupin Pharmaceuticals, Inc.
18.3.12.1. Company Overview
18.3.12.2. Financial Overview
18.3.12.3. Product Portfolio
18.3.12.4. Business Strategies
18.3.12.5. Recent Developments
18.3.13. Ocular Therapeutix, Inc.
18.3.13.1. Company Overview
18.3.13.2. Financial Overview
18.3.13.3. Product Portfolio
18.3.13.4. Business Strategies
18.3.13.5. Recent Developments
18.3.14. Novaliq GmbH
18.3.14.1. Company Overview
18.3.14.2. Financial Overview
18.3.14.3. Product Portfolio
18.3.14.4. Business Strategies
18.3.14.5. Recent Developments
18.3.15. ZEXUS PHARMA
18.3.15.1. Company Overview
18.3.15.2. Financial Overview
18.3.15.3. Product Portfolio
18.3.15.4. Business Strategies
18.3.15.5. Recent Developments
18.3.16. Grevis Pharmaceuticals Pvt. Ltd.
18.3.16.1. Company Overview
18.3.16.2. Financial Overview
18.3.16.3. Product Portfolio
18.3.16.4. Business Strategies
18.3.16.5. Recent Developments
List of Tables
Table 01: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Drug Type, 2020 to 2035
Table 02: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Anti-VEGF Drugs, 2020 to 2035
Table 03: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Route of Administration, 2020 to 2035
Table 04: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Dosage Form, 2020 to 2035
Table 05: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Product Type, 2020 to 2035
Table 06: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Indication, 2020 to 2035
Table 07: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Distribution Channel, 2020 to 2035
Table 08: Global Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Region, 2020 to 2035
Table 09: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, by Country, 2020 to 2035
Table 10: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Drug Type, 2020 to 2035
Table 11: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Anti-VEGF Drugs, 2020 to 2035
Table 12: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Route of Administration, 2020 to 2035
Table 13: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Dosage Form, 2020 to 2035
Table 14: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Product Type, 2020 to 2035
Table 15: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Indication, 2020 to 2035
Table 16: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Distribution Channel, 2020 to 2035
Table 17: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, by Country / Sub-region, 2020 to 2035
Table 18: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Drug Type, 2020 to 2035
Table 19: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Anti-VEGF Drugs, 2020 to 2035
Table 20: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Route of Administration, 2020 to 2035
Table 21: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Dosage Form, 2020 to 2035
Table 22: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Product Type, 2020 to 2035
Table 23: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Indication, 2020 to 2035
Table 24: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Distribution Channel, 2020 to 2035
Table 25: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, by Country / Sub-region, 2020 to 2035
Table 26: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Drug Type, 2020 to 2035
Table 27: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Anti-VEGF Drugs, 2020 to 2035
Table 28: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Route of Administration, 2020 to 2035
Table 29: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Dosage Form, 2020 to 2035
Table 30: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Product Type, 2020 to 2035
Table 31: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Indication, 2020 to 2035
Table 32: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Distribution Channel, 2020 to 2035
Table 33: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, by Country / Sub-region, 2020 to 2035
Table 34: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Drug Type, 2020 to 2035
Table 35: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Anti-VEGF Drugs, 2020 to 2035
Table 36: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Route of Administration, 2020 to 2035
Table 37: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Dosage Form, 2020 to 2035
Table 38: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Product Type, 2020 to 2035
Table 39: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Indication, 2020 to 2035
Table 40: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Distribution Channel, 2020 to 2035
Table 41: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, by Country / Sub-region, 2020 to 2035
Table 42: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Drug Type, 2020 to 2035
Table 43: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Anti-VEGF Drugs, 2020 to 2035
Table 44: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Route of Administration, 2020 to 2035
Table 45: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Dosage Form, 2020 to 2035
Table 46: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Product Type, 2020 to 2035
Table 47: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Indication, 2020 to 2035
Table 48: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, By Distribution Channel, 2020 to 2035
List of Figures
Figure 01: Global Ophthalmic Drugs Market Value Share Analysis, By Drug Type, 2024 and 2035
Figure 02: Global Ophthalmic Drugs Market Attractiveness Analysis, By Drug Type, 2025 to 2035
Figure 03: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Anti-Allergy Drugs, 2020 to 2035
Figure 04: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Anti-infective Drugs, 2020 to 2035
Figure 05: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Nonsteroidal Drugs, 2020 to 2035
Figure 06: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Anti-VEGF Drugs, 2020 to 2035
Figure 07: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Gene Therapy Drugs, 2020 to 2035
Figure 08: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Others, 2020 to 2035
Figure 09: Global Ophthalmic Drugs Market Value Share Analysis, By Route of Administration, 2024 and 2035
Figure 10: Global Ophthalmic Drugs Market Attractiveness Analysis, By Route of Administration, 2025 to 2035
Figure 11: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Topical, 2020 to 2035
Figure 12: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Intraocular, 2020 to 2035
Figure 13: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Others, 2020 to 2035
Figure 14: Global Ophthalmic Drugs Market Value Share Analysis, By Dosage Form, 2024 and 2035
Figure 15: Global Ophthalmic Drugs Market Attractiveness Analysis, By Dosage Form, 2025 to 2035
Figure 16: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Gel, 2020 to 2035
Figure 17: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Drop, 2020 to 2035
Figure 18: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Ointment, 2020 to 2035
Figure 19: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Suspension & Solutions, 2020 to 2035
Figure 20: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Others, 2020 to 2035
Figure 21: Global Ophthalmic Drugs Market Value Share Analysis, By Product Type, 2024 and 2035
Figure 22: Global Ophthalmic Drugs Market Attractiveness Analysis, By Product Type, 2025 to 2035
Figure 23: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Prescription Drugs, 2020 to 2035
Figure 24: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Over-the-Counter Drugs, 2020 to 2035
Figure 25: Global Ophthalmic Drugs Market Value Share Analysis, By Indication, 2024 and 2035
Figure 26: Global Ophthalmic Drugs Market Attractiveness Analysis, By Indication, 2025 to 2035
Figure 27: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Glaucoma, 2020 to 2035
Figure 28: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Dry Eye Disease, 2020 to 2035
Figure 29: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Age-Related Macular Degeneration, 2020 to 2035
Figure 30: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Diabetic Retinopathy, 2020 to 2035
Figure 31: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Pink Eye, 2020 to 2035
Figure 32: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Cataracts, 2020 to 2035
Figure 33: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Retinal Detachment, 2020 to 2035
Figure 34: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Others, 2020 to 2035
Figure 35: Global Ophthalmic Drugs Market Value Share Analysis, By Distribution Channel, 2024 and 2035
Figure 36: Global Ophthalmic Drugs Market Attractiveness Analysis, By Distribution Channel, 2025 to 2035
Figure 37: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Hospital Pharmacies, 2020 to 2035
Figure 38: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Retail Pharmacies, 2020 to 2035
Figure 39: Global Ophthalmic Drugs Market Revenue (US$ Bn), by Online Pharmacies, 2020 to 2035
Figure 40: Global Ophthalmic Drugs Market Value Share Analysis, By Region, 2024 and 2035
Figure 41: Global Ophthalmic Drugs Market Attractiveness Analysis, By Region, 2025 to 2035
Figure 42: North America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, 2020 to 2035
Figure 43: North America - Ophthalmic Drugs Market Value Share Analysis, by Country, 2024 and 2035
Figure 44: North America - Ophthalmic Drugs Market Attractiveness Analysis, by Country, 2025 to 2035
Figure 45: North America - Ophthalmic Drugs Market Value Share Analysis, By Drug Type, 2024 and 2035
Figure 46: North America - Ophthalmic Drugs Market Attractiveness Analysis, By Drug Type, 2025 to 2035
Figure 47: North America - Ophthalmic Drugs Market Value Share Analysis, By Route of Administration, 2024 and 2035
Figure 48: North America - Ophthalmic Drugs Market Attractiveness Analysis, By Route of Administration, 2025 to 2035
Figure 49: North America - Ophthalmic Drugs Market Value Share Analysis, By Dosage Form, 2024 and 2035
Figure 50: North America - Ophthalmic Drugs Market Attractiveness Analysis, By Dosage Form, 2025 to 2035
Figure 51: North America - Ophthalmic Drugs Market Value Share Analysis, By Product Type, 2024 and 2035
Figure 52: North America - Ophthalmic Drugs Market Attractiveness Analysis, By Product Type, 2025 to 2035
Figure 53: North America - Ophthalmic Drugs Market Value Share Analysis, By Indication, 2024 and 2035
Figure 54: North America - Ophthalmic Drugs Market Attractiveness Analysis, By Indication, 2025 to 2035
Figure 55: North America - Ophthalmic Drugs Market Value Share Analysis, By Distribution Channel, 2024 and 2035
Figure 56: North America - Ophthalmic Drugs Market Attractiveness Analysis, By Distribution Channel, 2025 to 2035
Figure 60: Europe - Ophthalmic Drugs Market Value (US$ Bn) Forecast, 2020 to 2035
Figure 61: Europe - Ophthalmic Drugs Market Value Share Analysis, by Country / Sub-region, 2024 and 2035
Figure 62: Europe - Ophthalmic Drugs Market Attractiveness Analysis, by Country / Sub-region, 2025 to 2035
Figure 63: Europe - Ophthalmic Drugs Market Value Share Analysis, By Drug Type, 2024 and 2035
Figure 64: Europe - Ophthalmic Drugs Market Attractiveness Analysis, By Drug Type, 2025 to 2035
Figure 65: Europe - Ophthalmic Drugs Market Value Share Analysis, By Route of Administration, 2024 and 2035
Figure 66: Europe - Ophthalmic Drugs Market Attractiveness Analysis, By Route of Administration, 2025 to 2035
Figure 67: Europe - Ophthalmic Drugs Market Value Share Analysis, By Dosage Form, 2024 and 2035
Figure 68: Europe - Ophthalmic Drugs Market Attractiveness Analysis, By Dosage Form, 2025 to 2035
Figure 69: Europe - Ophthalmic Drugs Market Value Share Analysis, By Product Type, 2024 and 2035
Figure 70: Europe - Ophthalmic Drugs Market Attractiveness Analysis, By Product Type, 2025 to 2035
Figure 72: Europe - Ophthalmic Drugs Market Value Share Analysis, By Indication, 2024 and 2035
Figure 73: Europe - Ophthalmic Drugs Market Attractiveness Analysis, By Indication, 2025 to 2035
Figure 74: Europe - Ophthalmic Drugs Market Value Share Analysis, By Distribution Channel, 2024 and 2035
Figure 75: Europe - Ophthalmic Drugs Market Attractiveness Analysis, By Distribution Channel, 2025 to 2035
Figure 76: Asia Pacific - Ophthalmic Drugs Market Value (US$ Bn) Forecast, 2020 to 2035
Figure 77: Asia Pacific - Ophthalmic Drugs Market Value Share Analysis, by Country/Sub-region, 2024 and 2035
Figure 78: Asia Pacific - Ophthalmic Drugs Market Attractiveness Analysis, by Country/Sub-region, 2025 to 2035
Figure 79: Asia Pacific - Ophthalmic Drugs Market Value Share Analysis, By Drug Type, 2024 and 2035
Figure 80: Asia Pacific - Ophthalmic Drugs Market Attractiveness Analysis, By Drug Type, 2025 to 2035
Figure 81: Asia Pacific - Ophthalmic Drugs Market Value Share Analysis, By Route of Administration, 2024 and 2035
Figure 82: Asia Pacific - Ophthalmic Drugs Market Attractiveness Analysis, By Route of Administration, 2025 to 2035
Figure 83: Asia Pacific - Ophthalmic Drugs Market Value Share Analysis, By Dosage Form, 2024 and 2035
Figure 84: Asia Pacific - Ophthalmic Drugs Market Attractiveness Analysis, By Dosage Form, 2025 to 2035
Figure 85: Asia Pacific - Ophthalmic Drugs Market Value Share Analysis, By Product Type, 2024 and 2035
Figure 86: Asia Pacific - Ophthalmic Drugs Market Attractiveness Analysis, By Product Type, 2025 to 2035
Figure 87: Asia Pacific - Ophthalmic Drugs Market Value Share Analysis, By Indication, 2024 and 2035
Figure 88: Asia Pacific - Ophthalmic Drugs Market Attractiveness Analysis, By Indication, 2025 to 2035
Figure 89: Asia Pacific - Ophthalmic Drugs Market Value Share Analysis, By Distribution Channel, 2024 and 2035
Figure 90: Asia Pacific - Ophthalmic Drugs Market Attractiveness Analysis, By Distribution Channel, 2025 to 2035
Figure 91: Latin America - Ophthalmic Drugs Market Value (US$ Bn) Forecast, 2020 to 2035
Figure 92: Latin America - Ophthalmic Drugs Market Value Share Analysis, by Country / Sub-region, 2024 and 2035
Figure 93: Latin America - Ophthalmic Drugs Market Attractiveness Analysis, by Country / Sub-region, 2025 to 2035
Figure 94: Latin America - Ophthalmic Drugs Market Value Share Analysis, By Drug Type, 2024 and 2035
Figure 95: Latin America - Ophthalmic Drugs Market Attractiveness Analysis, By Drug Type, 2025 to 2035
Figure 96: Latin America - Ophthalmic Drugs Market Value Share Analysis, By Route of Administration, 2024 and 2035
Figure 97: Latin America - Ophthalmic Drugs Market Attractiveness Analysis, By Route of Administration, 2025 to 2035
Figure 98: Latin America - Ophthalmic Drugs Market Value Share Analysis, By Dosage Form, 2024 and 2035
Figure 99: Latin America - Ophthalmic Drugs Market Attractiveness Analysis, By Dosage Form, 2025 to 2035
Figure 100: Latin America - Ophthalmic Drugs Market Value Share Analysis, By Product Type, 2024 and 2035
Figure 101: Latin America - Ophthalmic Drugs Market Attractiveness Analysis, By Product Type, 2025 to 2035
Figure 102: Latin America - Ophthalmic Drugs Market Value Share Analysis, By Indication, 2024 and 2035
Figure 103: Latin America - Ophthalmic Drugs Market Attractiveness Analysis, By Indication, 2025 to 2035
Figure 104: Latin America - Ophthalmic Drugs Market Value Share Analysis, By Distribution Channel, 2024 and 2035
Figure 105: Latin America - Ophthalmic Drugs Market Attractiveness Analysis, By Distribution Channel, 2025 to 2035
Figure 106: Middle East & Africa - Ophthalmic Drugs Market Value (US$ Bn) Forecast, 2020 to 2035
Figure 107: Middle East & Africa - Ophthalmic Drugs Market Value Share Analysis, by Country / Sub-region, 2024 and 2035
Figure 108: Middle East & Africa - Ophthalmic Drugs Market Attractiveness Analysis, by Country / Sub-region, 2025 to 2035
Figure 109: Middle East & Africa - Ophthalmic Drugs Market Value Share Analysis, By Drug Type, 2024 and 2035
Figure 110: Middle East & Africa - Ophthalmic Drugs Market Attractiveness Analysis, By Drug Type, 2025 to 2035
Figure 111: Middle East & Africa - Ophthalmic Drugs Market Value Share Analysis, By Route of Administration, 2024 and 2035
Figure 112: Middle East & Africa - Ophthalmic Drugs Market Attractiveness Analysis, By Route of Administration, 2025 to 2035
Figure 113: Middle East & Africa - Ophthalmic Drugs Market Value Share Analysis, By Dosage Form, 2024 and 2035
Figure 114: Middle East & Africa - Ophthalmic Drugs Market Attractiveness Analysis, By Dosage Form, 2025 to 2035
Figure 115: Middle East & Africa - Ophthalmic Drugs Market Value Share Analysis, By Product Type, 2024 and 2035
Figure 116: Middle East & Africa - Ophthalmic Drugs Market Attractiveness Analysis, By Product Type, 2025 to 2035
Figure 117: Middle East & Africa - Ophthalmic Drugs Market Value Share Analysis, By Indication, 2024 and 2035
Figure 118: Middle East & Africa - Ophthalmic Drugs Market Attractiveness Analysis, By Indication, 2025 to 2035
Figure 119: Middle East & Africa - Ophthalmic Drugs Market Value Share Analysis, By Distribution Channel, 2024 and 2035
Figure 120: Middle East & Africa - Ophthalmic Drugs Market Attractiveness Analysis, By Distribution Channel, 2025 to 2035