Reports
Reports
Global Cardiac Rhythm Management (CRM) Devices Batteries Market: Snapshot
As the availability of safer and cheaper lithium batteries is increasing, the global market for cardiac rhythm management (CRM) devices batteries is likely to attain new heights. CRM devices are designed to improve the health of patients with either too fast or too slow heartbeats. These devices are placed in the chests of patients suffering from arrhythmias or irregular heart rhythms. When a patient suffers from arrhythmias for too long, his/her heart cannot pump enough blood to the body, resulting in emergency situations, and even death. As these devices are powered by batteries, the demand for these devices will reflect in increased demand for these batteries.
According to the findings of Transparency Market Research (TMR), the global market for CRM devices batteries was valued at US$397.3 mn in 2015. The market is expected to expand at a CAGR of 3.0% between 2015 and 2024, reaching US$518.4 mn towards the end of 2024.
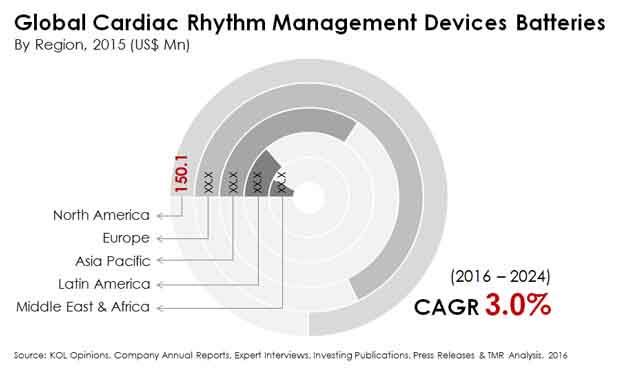
North America, Asia Pacific to Emerge as Key Markets
North America holds a strong position in the market presently, by virtue of its highly developed healthcare infrastructure and greater incidence of arrhythmias. The rising number of CRM implants is the prominent factor boosting the market for CRM devices batteries in this region. According to the findings of the U.S. CDC, nearly 750,000 hospitalizations each year in the U.S. are attributed to atrial fibrillation, the most common form of arrhythmia. The mortality rate associated with this condition was 130,000 deaths every year. The 2014 study also highlighted that as many as 2.7 to 6.1 million American citizens had atrial fibrillation. As a result, the number of CRM devices implants are slated to increase even further.
Asia Pacific is another region with great potential. The increasing healthcare expenditures of governments, growing per capita incomes of the middle class, and high prevalence of heart diseases, and growing awareness about heart diseases has been driving the market in Asia Pacific. Moreover, the development of highly efficient, technologically advanced CRM devices has been scooping out more demand. The increasing prevalence of cardiovascular diseases and sudden cardiac arrests will ensure that the demand for these products will continue to grow in China and India.
Technological Advancements to Ensure Greater Demand for ICDs and ICPs
Based on product type, the global market for CRM devices batteries has been segmented into cardiac resynchronization therapy devices, implantable cardiac pacemakers, and implantable cardioverter defibrillator. Implantable cardioverter defibrillator (ICD) is a device that records the heart rate of patients. The ICD is connected to the chambers of heart via two thin wires. As the heart beats, electric signals are sent to the ICD. As soon as an abnormal heartbeat is detected by this device, it aids the heart restore its normal pace. Cardiac defibrillation uses an electric shock to do this.
An implantable cardiac pacemaker (ICP), on the other hand, is a small device that assists the heart to produce more regular heartbeats with the help of a small electric stimulation. Both of these devices are typically placed under the skin of chest, below the collarbone. A cardiac resynchronization therapy device can be a half-dollar sized pacemaker that ‘resynchronizes’ the heart. As these devices improve the blood flow and efficiency of the heart, several heart patients suffering with moderate to severe symptoms are using these devices.
The efforts of ongoing research and development projects will come to fruition, producing next-generation CRM devices with efficient batteries. The batteries used in the aforementioned devices are expected to undergo significant transformations and upgrading, thereby accelerating demand.
Some of the major companies operating in the global market for CRM devices batteries are EaglePicher Technologies, Boston Scientific Corporation, BIOTRONIK SE & Co. KG, Greatbatch, Inc. (Integer), Quallion LLC, Medtronic, and St. Jude Medical, Inc.
High Statistics of Cardiac Arrhythmia makes North America leader in Cardiac Rhythm Management Devices Batteries Market
The surge in demand for cardiac rhythm management devices for patients suffering from irregular heart rhythms is creating vast demand for batteries for these devices. To meet this demand, the increasing availability of safer and cheaper lithium batteries that are suitable for cardiac rhythm management devices is serving to be useful. Meanwhile, cardiac rhythm management devices are designed to be placed in the chests of patients suffering from irregular heart rhythms. For individuals in this condition, the heart is unable to pump enough blood to the body, which could result in a medical emergency or even death.
North America is at the forefront among other key regions in the overall cardiac rhythm management devices batteries market. According to statistics of the U.S. CDC, almost 750,000 hospitalizations each year in the country are associated with atrial fibrillation, the most common form of arrhythmia, and mortality of 130,000 associated with this condition each year. Such high incidence of cardiac arrhythmia that needs to be managed using cardiac rhythm management devices is creating opportunities in the cardiac rhythm management devices batteries market of the region.
Asia Pacific is anticipated to display substantial demand for cardiac rhythm management devices in the future. Rising healthcare expenditures from governments in emerging economies of the region, rising per capita income of the middle class population, and increasing prevalence of heart diseases is creating the demand for both invasive and non-invasive cardiac procedure. This includes placing cardiac rhythm management devices, which, in turn, spells demand for cardiac rhythm management devices batteries. Furthermore, with the modernization of healthcare infrastructure in emerging economies of the region, the easy availability of highly advanced cardiac rhythm management devices is spelling growth in the cardiac rhythm management devices batteries market of the region.
Key product segments of the cardiac rhythm management devices batteries market include implantable cardioverter defibrillator, cardiac resynchronization therapy devices, and implantable cardiac pacemakers.
1. Preface
1.1. Market Definition and Scope
1.2. Market Segmentation
1.3. Key Research Objectives
1.4. Research Highlights
2. Assumptions and Research Methodology
3. Executive Summary : Global Cardiac Rhythm Management Devices Batteries Market
4. Market Overview
4.1. Introduction
4.1.1. Product Type Definition
4.2. Overview
4.3. Key Market Indicators
4.3.1. Region/Country Specific
4.3.2. Product/Application/Technology Specific
4.3.3. Target Market Specific – Global Scenario
4.4. Market Dynamics
4.4.1. Drivers
4.4.2. Restraints
4.4.3. Opportunity
4.5. Global Cardiac Rhythm Management Devices Batteries Market Analysis and Forecasts, 2015–2024
4.5.1. Market Volume Projections, 2015–2024
4.5.2. Pricing - Actuals and Projections, 2015–2024
4.5.3. Market Revenue Projections (US$ Mn)
4.6. Cardiac Rhythm Management Devices Batteries Market - Global Supply Demand Scenario
4.7. Porter’s Five Force Analysis
4.8. Value Chain Analysis
4.9. Market Outlook
4.10. Regulatory Scenario
4.11. Porter's Analysis
4.12. Indictions and Recommendations
5. Global Cardiac Rhythm Management Devices Batteries Market Analysis and Forecasts, By Product Type
5.1. Introduction & Definition
5.2. Key Findings / Developments
5.3. Key Trends
5.4. Market Value and Volume Forecast By Product Type , 2015–2024
5.4.1. Implantable Cardiac Pacemakers
5.4.2. Implantable Cardioverter Defibrillators
5.5. Product Type Comparison Matrix
5.6. Market Attractiveness By Product Type
6. Global Cardiac Rhythm Management Devices Batteries Market Analysis and Forecasts, By Power Source
6.1. Introduction & Definition
6.2. Key Findings / Developments
6.3. Key Trends
6.4. Market Value and Volume Forecast By Power Source , 2015–2024
6.4.1. Lithium-iodine Cells
6.4.2. Lithium-silver Vanadium Oxide Cell
6.4.3. Lithium-carbon Mono-fluoride Cells
6.4.4. Lithium-manganese Dioxide Cells
6.5. Power Source Comparison Matrix
6.6. Market Attractiveness By Power Source
7. Global Cardiac Rhythm Management Devices Batteries Market Analysis and Forecasts, By Region
7.1. Key Findings
7.2. Policies and Regulations
7.3. Market Value and Volume Forecast By Region
7.3.1. North America
7.3.2. Latin America
7.3.3. Europe
7.3.4. Asia Pacific
7.3.5. MEA
7.4. Market Attractiveness By Country/Region
8. North America Cardiac Rhythm Management Devices Batteries Market Analysis and Forecast
8.1. Introduction
8.1.1. Key Findings
8.1.2. Policies and Regulations
8.1.3. Price Trend Analysis
8.1.4. Key Trends
8.2. Market Value and Volume Forecast By Product Type , 2015–2024
8.2.1. Implantable Cardiac Pacemakers
8.2.2. Implantable Cardioverter Defibrillators
8.3. Market Value and Volume Forecast By Power Source , 2015–2024
8.3.1. Lithium-iodine Cells
8.3.2. Lithium-silver Vanadium Oxide Cell
8.3.3. Lithium-carbon Mono-fluoride Cells
8.3.4. Lithium-manganese Dioxide Cells
8.4. Market Value and Volume Forecast By Country , 2015–2024
8.4.1. The U.S.
8.4.2. Canada
8.5. Market Attractiveness Analysis
8.5.1. By Product Type
8.5.2. By Power Source
8.5.3. By Country
9. Latin America Cardiac Rhythm Management Devices Batteries Market Analysis and Forecast
9.1. Introduction
9.1.1. Key Findings
9.1.2. Policies and Regulations
9.1.3. Price Trend Analysis
9.1.4. Key Trends
9.2. Market Value and Volume Forecast By Product Type , 2015–2024
9.2.1. Implantable Cardiac Pacemakers
9.2.2. Implantable Cardioverter Defibrillators
9.3. Market Value and Volume Forecast By Power Source , 2015–2024
9.3.1. Lithium-iodine Cells
9.3.2. Lithium-silver Vanadium Oxide Cell
9.3.3. Lithium-carbon Mono-fluoride Cells
9.3.4. Lithium-manganese Dioxide Cells
9.4. Market Value and Volume Forecast By Country , 2015–2024
9.4.1. Brazil
9.4.2. Mexico
9.4.3. Rest of Latin America
9.5. Market Attractiveness Analysis
9.5.1. By Product Type
9.5.2. By Power Source
9.5.3. By Country
10. Europe Cardiac Rhythm Management Devices Batteries Market Analysis and Forecast
10.1. Introduction
10.1.1. Key Findings
10.1.2. Policies and Regulations
10.1.3. Price Trend Analysis
10.1.4. Key Trends
10.2. Market Value and Volume Forecast By Product Type , 2015–2024
10.2.1. Implantable Cardiac Pacemakers
10.2.2. Implantable Cardioverter Defibrillators
10.3. Market Value and Volume Forecast By Power Source , 2015–2024
10.3.1. Lithium-iodine Cells
10.3.2. Lithium-silver Vanadium Oxide Cell
10.3.3. Lithium-carbon Mono-fluoride Cells
10.3.4. Lithium-manganese Dioxide Cells
10.4. Market Value and Volume Forecast By Country , 2015–2024
10.4.1. Germany
10.4.2. France
10.4.3. The U.K.
10.4.4. Italy
10.4.5. Spain
10.4.6. Rest of Europe
10.5. Market Attractiveness Analysis
10.5.1. By Product Type
10.5.2. By Power Source
10.5.3. By Country
11. Asia Pacific Cardiac Rhythm Management Devices Batteries Market Analysis and Forecast
11.1. Introduction
11.1.1. Key Findings
11.1.2. Policies and Regulations
11.1.3. Price Trend Analysis
11.1.4. Key Trends
11.2. Market Value and Volume Forecast By Product Type , 2015–2024
11.2.1. Implantable Cardiac Pacemakers
11.2.2. Implantable Cardioverter Defibrillators
11.3. Market Value and Volume Forecast By Power Source , 2015–2024
11.3.1. Lithium-iodine Cells
11.3.2. Lithium-silver Vanadium Oxide Cell
11.3.3. Lithium-carbon Mono-fluoride Cells
11.3.4. Lithium-manganese Dioxide Cells
11.4. Market Value and Volume Forecast By Country , 2015–2024
11.4.1. China
11.4.2. Japan
11.4.3. India
11.4.4. Australia & New Zealand
11.4.5. Rest of APAC
11.5. Market Attractiveness Analysis
11.5.1. By Product Type
11.5.2. By Power Source
11.5.3. By Country
12. MEA Cardiac Rhythm Management Devices Batteries Market Analysis and Forecast
12.1. Introduction
12.1.1. Key Findings
12.1.2. Policies and Regulations
12.1.3. Price Trend Analysis
12.1.4. Key Trends
12.2. Market Value and Volume Forecast By Product Type , 2015–2024
12.2.1. Implantable Cardiac Pacemakers
12.2.2. Implantable Cardioverter Defibrillators
12.3. Market Value and Volume Forecast By Power Source , 2015–2024
12.3.1. Lithium-iodine Cells
12.3.2. Lithium-silver Vanadium Oxide Cell
12.3.3. Lithium-carbon Mono-fluoride Cells
12.3.4. Lithium-manganese Dioxide Cells
12.4. Market Value and Volume Forecast By Country , 2015–2024
12.4.1. GCC Countries
12.4.2. South Africa
12.4.3. Rest of MEA
12.5. Market Attractiveness Analysis
12.5.1. By Product Type
12.5.2. By Power Source
12.5.3. By Country
13. Competition Landscape
13.1. Market Player – Competition Matrix (By Tier and Size of companies)
13.2. Market Share Analysis By Company (2015)
13.3. Company Profiles (Details – Overview, Financials, Recent Developments, Strategy)
13.3.1. Medtronic
13.3.1.1. Company Overview (HQ, Business Segments, Employee Strength)
13.3.1.2. Financial Overview
13.3.1.3. Product Portfolio
13.3.1.4. SWOT Analysis
13.3.1.5. Strategic Overview
13.3.2. Boston Scientific Corporation
13.3.2.1. Company Overview (HQ, Business Segments, Employee Strength)
13.3.2.2. Financial Overview
13.3.2.3. Product Portfolio
13.3.2.4. SWOT Analysis
13.3.2.5. Strategic Overview
13.3.3. St. Jude Medical, Inc.
13.3.3.1. Company Overview (HQ, Business Segments, Employee Strength)
13.3.3.2. Financial Overview
13.3.3.3. Product Portfolio
13.3.3.4. SWOT Analysis
13.3.3.5. Strategic Overview
13.3.4. Greatbatch, Inc. (Integer)
13.3.4.1. Company Overview (HQ, Business Segments, Employee Strength)
13.3.4.2. Financial Overview
13.3.4.3. Product Portfolio
13.3.4.4. SWOT Analysis
13.3.4.5. Strategic Overview
13.3.5. EaglePicher Technologies, LLC
13.3.5.1. Company Overview (HQ, Business Segments, Employee Strength)
13.3.5.2. Financial Overview
13.3.5.3. Product Portfolio
13.3.5.4. SWOT Analysis
13.3.5.5. Strategic Overview
13.3.6. Quallion LLC
13.3.6.1. Company Overview (HQ, Business Segments, Employee Strength)
13.3.6.2. Financial Overview
13.3.6.3. Product Portfolio
13.3.6.4. SWOT Analysis
13.3.6.5. Strategic Overview
13.3.7. BIOTRONIK SE & Co. KG
13.3.7.1. Company Overview (HQ, Business Segments, Employee Strength)
13.3.7.2. Financial Overview
13.3.7.3. Product Portfolio
13.3.7.4. SWOT Analysis
13.3.7.5. Strategic Overview
List of Tables
Table 01: Global Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Product Type, 2016–2024
Table 02: Global Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Product Type, 2016–2024
Table 03: Global Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Power Source, 2016–2024
Table 04: Global Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Power Source, 2016–2024
Table 05: Global Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Region, 2016–2024
Table 06: Global Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Region, 2016–2024
Table 07: North America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Product Type, 2016–2024
Table 08: North America Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Product Type, 2016–2024
Table 09: North America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Power Source, 2016–2024
Table 10: North America Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Power Source, 2016–2024
Table 11: North America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Country, 2016–2024
Table 12: Latin America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Product Type, 2016–2024
Table 13: Latin America Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Product Type, 2016–2024
Table 14: Latin America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Power Source, 2016–2024
Table 15: Latin America Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Power Source, 2016–2024
Table 16: Latin America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Country, 2016–2024
Table 17: Europe Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Product Type, 2016–2024
Table 18: Europe Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Product Type, 2016–2024
Table 19: Europe Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Power Source, 2016–2024
Table 20: Europe Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Power Source, 2016–2024
Table 21: Europe Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Country, 2016–2024
Table 22: Asia Pacific Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Product Type, 2016–2024
Table 23: Asia Pacific Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Product Type, 2016–2024
Table 24: Asia Pacific Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Power Source, 2016–2024
Table 25: Asia Pacific Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Power Source, 2016–2024
Table 26: Asia Pacific Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Country, 2016–2024
Table 27: Middle East & Africa Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Product Type, 2016–2024
Table 28: Middle East & Africa Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Product Type, 2016–2024
Table 29: Middle East & Africa Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Power Source, 2016–2024
Table 30: Middle East & Africa Cardiac Rhythm Management Devices Batteries Volume (Mn Units) Forecast, by Power Source, 2016–2024
Table 31: Middle East & Africa Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, by Country, 2016–2024
List Of Figures
Figure 01: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) & Growth (%) Forecast, 2016–2024
Figure 02: Global Cardiac Rhythm Management Devices Batteries Market Value Share, by Product Type (2016)
Figure 03: Global Cardiac Rhythm Management Devices Batteries Market Value Share, by Power Source (2016)
Figure 04: Global Cardiac Rhythm Management Devices Batteries Market Value Share, by Region (2016)
Figure 05: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) Share Analysis, by Product Type, 2016 and 2024
Figure 06: Global Implantable Cardiac Pacemakers Market Value Forecast, 2016–2026 and Y-o-Y Growth Projection, 2015–2024
Figure 07: Global Implantable Cardioverter Defibrillators Market Value Forecast, 2016–2026 and Y-o-Y Growth Projection, 2015–2024
Figure 08: Global Cardiac Resynchronization Therapy Devices Market Value Forecast, 2016–2026 and Y-o-Y Growth Projection, 2015–2024
Figure 09: Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Product Type, 2016–2024
Figure 10: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) Share Analysis, by Power Source, 2016 and 2024
Figure 11: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) & Growth (%) Forecast, by Lithium-iodide Cells Segment, 2016–2024
Figure 12: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) & Growth (%) Forecast, by Lithium-silver Vanadium Oxide Cells Segment, 2016–2024
Figure 13: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) & Growth (%) Forecast, by Lithium-carbon Mono-fluoride Cells Segment, 2016–2024
Figure 14: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) & Growth (%) Forecast, by Lithium- manganese Dioxide Cells Segment, 2016–2024
Figure 15: Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Power Source, 2016–2024
Figure 16: Global Cardiac Rhythm Management Devices Batteries Market Value (US$ Mn) Share Analysis, by Region, 2016 and 2024
Figure 17: Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Region, 2016–2024
Figure 18: North America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, 2016–2024
Figure 19: North America Cardiac Rhythm Management Devices Batteries Market Size Y-o-Y Growth Projections, 2016–2024
Figure 20: North America Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Product Type, 2016–2024
Figure 21: North America Orthopedic Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Power Source, 2016–2024
Figure 22: North America Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Country, 2016–2024
Figure 23: Latin America Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, 2016–2024
Figure 24: Latin America Cardiac Rhythm Management Devices Batteries Market Size Y-o-Y Growth Projections, 2016–2024
Figure 25: Latin America Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Product Type, 2016–2024
Figure 26: Latin America Orthopedic Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Power Source, 2016–2024
Figure 27: Latin America Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Country, 2016–2024
Figure 28: Europe Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, 2016–2024
Figure 29: Europe Cardiac Rhythm Management Devices Batteries Market Size Y-o-Y Growth Projections, 2016–2024
Figure 30: Europe Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Product Type, 2016–2024
Figure 31: Europe Orthopedic Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Power Source, 2016–2024
Figure 32: Europe Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Country, 2016–2024
Figure 33: Asia Pacific Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, 2016–2024
Figure 34: Asia Pacific Cardiac Rhythm Management Devices Batteries Market Size Y-o-Y Growth Projections, 2016–2024
Figure 35: Asia Pacific Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Product Type, 2016–2024
Figure 36: Asia Pacific Orthopedic Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Power Source, 2016–2024
Figure 37: Asia Pacific Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Country, 2016–2024
Figure 38: Middle East & Africa Cardiac Rhythm Management Devices Batteries Market Size (US$ Mn) Forecast, 2016–2024
Figure 39: Middle East & Africa Cardiac Rhythm Management Devices Batteries Market Size Y-o-Y Growth Projections, 2016–2024
Figure 40: Middle East & Africa Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Product Type, 2016–2024
Figure 41: Middle East & Africa Orthopedic Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Power Source, 2016–2024
Figure 42: Middle East & Africa Cardiac Rhythm Management Devices Batteries Market Attractiveness Analysis, by Country, 2016–2024